- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
ICMR Study Reveals, Covid Vaccines NOT Behind Sudden Deaths—Flags Other Risk Factors
Credits: Canva
As Covid-19 vaccination campaigns reached billions worldwide, so too did widespread public fear of infrequent but highly publicized cases of sudden death after vaccination. Over the past few months, the fear has sparked a spate of conjecture and disinformation, particularly on social media. On Wednesday, the Government of India's Ministry of Health and Family Welfare (MoHFW) released a statement of the utmost certainty: there is no causative association between Covid-19 vaccines and sudden death in adults. This finding, based on wide-ranging research conducted by the All India Institute of Medical Sciences (AIIMS) and the Indian Council of Medical Research (ICMR), is a watershed event for public health worldwide.
The MoHFW stands on the basis of two large, methodologically sound studies. The first, by ICMR's National Institute of Epidemiology, was a multicentric case–control study carried out over 47 Indian hospitals between May and August 2023. The second, a prospective study currently being conducted by AIIMS in association with ICMR, is investigating causes of sudden unexpected deaths among young adults.
What Prompted the Investigation?
In the past year, increased media coverage and public anxiety about sudden, unexpected deaths, especially among those aged 18–45, fueled a spate of misinformation that some tried to attribute to Covid-19 vaccination. In response, the Indian government launched extensive investigations in various institutions to give a data-based analysis of these incidents.
The health ministry emphasized that some of the national agencies, such as ICMR, AIIMS, and the National Centre for Disease Control (NCDC), carried out systematic studies to investigate the underlying reasons behind sudden deaths among adults. The objective was to conclude with certainty whether or not there exists any relation between the said deaths and earlier Covid-19 vaccinations.
A multicentre matched case–control study, "Factors associated with unexplained sudden deaths among adults aged 18–45 years in India", was carried out by ICMR's National Institute of Epidemiology during May-August 2023. This study covered 47 hospitals and aimed at determining the potential correlation between Covid-19 vaccination and unexplained deaths in young adults.
The verdict was clear: no heightened risk of sudden death from Covid-19 vaccines.
At the same time, AIIMS is carrying out a long-term prospective study named "Establishing the cause in sudden unexplained deaths in young," which is funded and supported by ICMR. The preliminary results support ICMR's findings. The top causes of sudden death remain:
- Myocardial infarction (heart attack)
- Genetic predispositions (inherited heart rhythm disorders)
- Lifestyle issues, including alcohol misuse, drug abuse, smoking, and overexertion
- Post-Covid complications like cardiac inflammation or clotting abnormalities
Interestingly, scientists noted that the cause patterns have continued to follow trends similar to those observed in history. That is, the rates of sudden deaths in young adults have not presented any significant upward leap or shift that would be traceable to the Covid-19 vaccination campaign.
Debunking Misinformation
The health ministry’s statement comes amid a surge of unfounded claims linking Covid-19 vaccines to heart attacks and sudden deaths. “Scientific experts have reiterated that statements linking Covid vaccination to sudden deaths are false and misleading, and are not supported by scientific consensus,” the ministry said. Such speculation, they warn, can erode public trust in vaccines and fuel hesitancy, potentially reversing the hard-won gains of global immunization campaigns.
Union Minister of Health Mansukh Mandaviya highlighted the role of evidence-based decision-making: "The ICMR report yielded strong evidence discrediting insinuations that the vaccines were behind heart attacks or sudden death. Decisions have to be made on scientific studies and facts, not on misinformation."
Vaccines Remain Safe and Essential
The Health Ministry reaffirmed that Covid-19 vaccines given in India are safe and effective, and cases of serious adverse events are very rare. The statement highlighted that the vaccines cannot be blamed for sudden death, particularly without scientific evidence.
The ministry claims that disinformation campaigns attributing vaccines to deaths can be harmful. "Scientific experts have reaffirmed that claims associating Covid vaccination with sudden death are untrue and misleading," the official release said. "Speculative reports in the absence of conclusive evidence may undermine public trust in vaccines, which have saved millions of lives."
What Is Sudden Death?
Sudden death is a sudden and rapid cardiac arrest in people who typically are otherwise healthy. Since it happens unexpectedly, there is generally no way to prevent or even anticipate it. Without prompt emergency medical treatment, sudden cardiac arrest invariably leads to fatality.
Identifying Symptoms of Sudden Death
Sudden death normally happens without warning symptoms. In certain situations, though, there are a few warning signs that flash very briefly, such as:
- Near-fainting or dizziness
- Sudden loss of consciousness
- No response to outside stimuli
- Stoppage of breathing
- Skin becomes a bluish or purplish color
- Early detection and emergency treatment are essential, as time is of the essence.
What Causes Sudden Death?
The most common reason for sudden cardiac death is ventricular fibrillation—a life-threatening heart rhythm disorder (arrhythmia) that causes the heart to lose its ability to pump blood effectively. When the heart fails to provide a normal rhythm, blood flow to the body's vital organs, including the brain, is interrupted, rapidly becoming fatal.
Dr Amit Bhushan Sharma, Director & Unit Head, Cardiologist, Paras Health, Gurugram explains, "The recent findings by ICMR and NCDC are both timely and reassuring, clearly showing no direct link between COVID-19 vaccines and sudden cardiac deaths. In most such cases, the root causes are underlying health conditions like undiagnosed coronary artery disease, inherited heart disorders, or lifestyle factors such as chronic stress, obesity, smoking, and physical inactivity. Cardiac arrest can occur suddenly, but there are often prior signs of cardiac issues—such as chest pain or discomfort, shortness of breath, persistent fatigue, heart palpitations, or episodes of fainting—which tend to be gradual and progressive."
"On the other hand, the common post-vaccine symptoms—like mild fever, fatigue, muscle aches, or arm soreness—are short-term and self-limiting, with no impact on the heart's electrical or structural functioning. These vaccine-related side effects are completely different from the symptoms or risk patterns seen in cardiac arrest cases. It’s crucial that we focus on heart health through regular screenings and lifestyle management, and continue to trust vaccines, which have been proven safe and essential in preventing severe COVID-19 outcomes."
Underlying Heart Conditions Associated with Sudden Death
Certain underlying diseases of the heart may predispose a person to sudden cardiac arrest:
Hypertrophic Cardiomyopathy: A genetic condition characterized by the thickening of the heart muscle, typically the left ventricle. It is the leading cause of sudden death in adults under age 50.
Dilated Cardiomyopathy: Characterized by enlargement and reduced pumping function of the left ventricle. Although most cases are not clearly caused by a specific disease, viral infections are sometimes responsible.
Arrhythmogenic Right Ventricular Dysplasia: A genetic disorder in which heart muscle cells are progressively replaced with fatty tissue, primarily found in adults younger than 40.
Brugada Syndrome: An inherited disorder that results in potentially fatal attacks of rapid, irregular heart rhythms (ventricular tachycardia), causing fainting or sudden death.
Long QT Syndrome: A disorder characterized by structural abnormalities in the heart's sodium and potassium channels, causing erratic heart rhythms.
Catecholaminergic Polymorphic Ventricular Tachycardia: An inherited condition in which the heart is unusually sensitive to adrenaline, making it more likely to develop potentially fatal arrhythmias.
COVID Cicada variant: Are Children More At Risk? How To Safeguard?

Credit: iStock
The COVID Cicada variant, currently circulating in 23 countries, is likely to affect children more than the previous variants.
Children have largely escaped severe illness from the SARS-COV-2 virus.
However, the new Cicada variant with around 75 genetic changes in its spike protein is likely to disproportionately affect children, as per an expert, who noted its presence in the UK.
“Some people have done analysis on this, suggesting it may be more prevalent among young children. Children get infections all the time, but this might be something to do with the fact that they have never been exposed to Covid vaccines," Prof Ravindra Gupta, of Cambridge University, who advised the UK government during the pandemic, was quoted as saying to The Mirror.
“So this is something we’re looking at in the lab to try and work out why. The problem with this is that it is an infection that spreads fast. Eventually, it ends up in someone vulnerable," he added.
Key Symptoms
Symptoms seem to be similar to those of other recent variants and include
- sore throat,
- cough,
- congestion,
- fatigue,
- headache
- fever.
Also read: Cicada Variant: Will The Current COVID Vaccine Provide Protection?
What Do The 75 Genetic Changes Mean?
Cicada, a descendant of the Omicron variant that emerged in 2021, was first detected in South Africa in 2024. However, it disappeared soon after, only to emerge early this year.
Prof Gupta told the publication that “the Cicada variant developed the 75 mutations to escape immunity, and it evolved by incubating a single patient for over a year”.
"This 'patient zero' would have been unable to clear the virus due to a compromised immune system," due to HIV or anti-cancer drugs.
Further, he explained that due to 75 mutations to the spike protein, the body’s antibodies will take a longer time to fight the Cicada variant.
However, there is no evidence yet that it is likely to cause more severe disease.
Will The Cicada Variant Cause Severe Illness?
The Cicada variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
Yet, the World Health Organization and health experts advise that existing COVID vaccinations can help prevent severe illness and hospitalization.
"It would appear that all the protections we have from our experience with the virus and with vaccines probably offer more limited—not zero—but more limited protection against this strain," Dr. William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center, was quoted as saying by Time.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
How To Safeguard
Dr. Sai Balasubramanian, a doctor and healthcare strategy executive, writing in Forbes, stressed the need to follow COVID practices such as masks and hand hygiene.
"Healthcare professionals recommend taking general precautions, similar to those used to prevent most viral transmission: get vaccinations when appropriate, wear masks in crowded areas or indoors where there is a high risk of transmission," he said.
He also urged “avoid individuals who have known illness or infections, wash hands frequently, and continue to stay apprised of local community guidelines and the infection spread”.
India To Strictly Inspect GLP-1 Drugs To Curb Misuse: Govt Flags Risks Amid Weight-loss Hype

Credit: iStock
GLP-1 drugs are powerful tools that can improve health, but it comes with risks and caveats. While these are medical therapies, they are being misused as cosmetic products by cosmetologists, physiotherapists, dermatologists, among others.
To curb its misuse and improve public health, the Indian government today issued guidelines on its use, while also flagging risks around it.
In an official statement issued today, the government has stated that it will roll out stricter inspections to curb the misuse of GLP-1 drugs in the country and will punish those who violate the rules.
“GLP-1 drugs are a significant medical breakthrough in treating type 2 diabetes and obesity, but they are not without risk. The drugs carry a wide range of side effects - from common symptoms like nausea and vomiting to severe complications including pancreatitis, kidney injury, and bowel obstruction. These risks make it essential that GLP-1 drugs are only taken under the supervision of registered medical practitioners/specialists,” the statement said.
In the wake of its hype over weight loss, the country's regulatory authorities have taken firm steps to prevent unsupervised use and malpractices in the supply chain.
It has “strongly advised” patients and the public to” consult a qualified medical specialist before use, and to obtain these medications with prescription only through legitimate, regulated channels”.
“Stricter inspections and surveillance will continue in the coming weeks. Businesses that break the rules will face license cancellation, fines, and legal action,” the statement said.
Also read: GLP-1 Drugs: Why India Needs Stricter Rules Now
What Are The Side-effects Of GLP-1 Drugs?
GLP-1 drugs work to keep food in the digestive system for longer – making people feel fuller for longer, thus reducing appetite and inducing weight loss. Hence, the drugs are also prescribed to people with obesity.
However, when done without clinical oversight, it can lead to severe health complications.
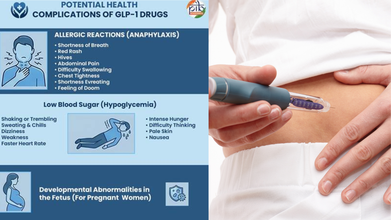
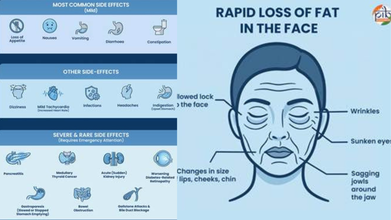
There are various side effects to taking GLP-1 drugs, including nausea and dizziness, to pancreatitis and medullary thyroid cancer.
Some serious side effects include
- Pancreatis
- Thyroid Cancer
- Acute (Sudden) Kidney Injury
- Worsening Diabetes-Related Retinopathy
- Gallstone & Bile Duct Blockage
- Rapid loss of fat in the face
- Wrinkles
- Sunken eyes
GLP-1 medications may also complicate various health conditions, such as allergic reactions, with shortness of breath and low blood sugar.
Rush For Generic Weight Loss Drugs
The government's advisory comes as patent for semaglutide -- an active ingredient in diabetes and anti-obesity drugs, specifically Wegovy and Ozempic -- expired on March 20.
This has led to a rush among Indian pharma companies to launch cheaper generic versions, significantly increasing affordability and access for millions battling Type 2 diabetes and obesity.
Also read: CDSCO threatens action against pharma companies for promoting GLP-1 weight-loss drugs
Major Indian drugmakers who launched their generic semglutide injection in the country in March include Sun Pharmaceutical Industries, Zydus Lifesciences, Alkem Laboratories, and Dr. Reddy’s Laboratories.
WHO Guidelines on GLP-1 drugs
Amid the increasing prevalence of GLP-1 drugs, the World Health Organisation (WHO), late last year, acknowledged its role in treating obesity.
However, it warned that medications like GLP-1 alone will not solve the problem affecting more than one billion people worldwide.
The global health body also issued conditional recommendations for using these therapies as part of a comprehensive approach that includes healthy diets, regular physical activity, and support from health professionals.
Ozempic And Wegovy Prices Slashed Again In India To Counter Cheaper Generics

Credit: iStock
Danish pharma giant Novo Nordisk has once again reduced the prices of its blockbuster drugs Ozempic and Wegovy in India. The strategic move is aimed at staying ahead of the entry of the low-cost generic competition in the country.
Starting today, the Ozempic price in India will be cut by 36 percent, while Wegovy has seen a steeper 48 percent reduction — limiting the base price below Rs 6,000 per month.
While Ozempic is available in three dose strengths of 0.25 mg, 0.5 mg, and 1 mg in India, Wegovy has five dose strengths.
Ozempic's and Wegovy's lowest doses of 0.25 mg will now be priced at Rs 1,415 for a weekly shot from Rs 2,200 and Rs 2,712 earlier, respectively, Novo Nordisk India said in a statement on Tuesday.
The average price reduction across doses is 23.8 percent for Ozempic and 27 percent for Wegovy, it added.
Speaking to the media, Novo Nordisk stated that the price cut will benefit a large section of people with diabetes and obesity in India.
The generic weight loss drugs will open up a huge customer base, especially in India, with a high burden of diabetes and obesity.
Novo first slashed Wegovy's price for the first time by up to 37 percent from its launch price last year.
In addition to the entry of generics, the launch of rival Eli Lilly's blockbuster diabetes and obesity drug Mounjaro in India in 2025.
Also read: Semaglutide Becomes Cheap In India: A Gamechanger Or Health Gamble?
Entry Of Generics In India
Soon after the end of Novo Nordisk's patent on semaglutide, on March 20, a host of companies in India launched generic versions of Ozempic.
This includes NATCO Pharma's Semanat and Semafull, priced at Rs 1,290 per month and Rs 1,750 — about 90 per cent cheaper than Ozempic, costing Rs 8,800 per month.
Eris Lifesciences also announced its plans to launch a multi-dose vial under the brand name Sundae at the same price.
Dr. Reddy’s Laboratories, Sun Pharmaceutical Industries, and Glenmark Pharmaceuticals have also launched their generic versions of semaglutide.
Dr. Reddy’s Obeda is priced at Rs 4,200 per month, available in 2 mg and 4 mg strengths.
Sun Pharmaceutical Industries will sell under the brand names Noveltreat (doses ranging from Rs 900) and Sematrinity (doses ranging from Rs 750). Meanwhile, Glenmark’s GLIPIQ vials range from Rs 325 to Rs 440 per week.
Zydus Lifesciences has also announced the launch of its generic version of semaglutide injection under the brand names SEMAGLYNTM, MASHEMATM, and ALTERMET. The average monthly cost of the treatment will be about Rs 2,200, the company said in a statement.
How Does Semaglutide Work?
Semaglutide works as a GLP-1 receptor agonist that mimics the GLP-1 hormone to regulate appetite and blood sugar. It slows gastric emptying and makes you feel fuller longer. It also signals the brain to reduce hunger and cravings, and triggers the pancreas to release insulin when blood sugar is high.
Also read: This Pill Is More Effective For Weight Loss Than Ozempic, Scientists Say
They work by increasing insulin release in a glucose-dependent manner, decreasing the liver's production of glucagon, and slowing down the emptying of the stomach, which helps lower blood sugar levels after a meal. They also act on the brain to suppress appetite and increase feelings of fullness, leading to reduced calorie intake.
In people with type 2 diabetes, notes Harvard Health, the body's cells are resistant to the effects of insulin and the body does not produce enough insulin, or both. This is when GLP-1 agonists stimulate the pancreas to release insulin and suppress the release of another hormone called glucagon.
These drugs also act in the brain to reduce hunger and act on the stomach to delay emptying, so you feel full for a longer time. These effects can lead to weight loss, which can be an important part of managing diabetes.
© 2024 Bennett, Coleman & Company Limited

