- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Over 2 Million Eye Drops Recalled In U.S. Amid Alarming Risk Of Blindness And Irreversible Damage
Credits: Canva
A massive recall has been issued for over 2 million eye drop cartons due to concerns over sterility that can be potentially harmful and lead to serious eye infections, blindness, and even permanent blindness in extreme cases.
The voluntary recall, issued jointly by AvKARE, a Tennessee pharmaceutical distributor, and the U.S. Food and Drug Administration (FDA), is being labeled a Class II recall—meaning that although the danger of serious harm is relatively minor, the harm can be transient or medically reversible.
The eye drops were recalled from store shelves after an FDA audit uncovered manufacturing deficiencies that rendered product sterility in doubt. Although neither the FDA nor AvKARE would reveal the specifics regarding the manufacturing deficiencies, the notice of recall mentioned a "lack of assurance of sterility"—a rather disturbing red flag for products that are applied directly to the eyes.
Ophthalmic sterility is non-negotiable. A compromise of sterility, however slight, invites bacterial contamination, which has the potential to penetrate the soft tissues of the eye and develop catastrophic infections.
These products have been shipped between May 26, 2023, and April 21, 2025, which could have endangered millions of customers—particularly those who are addressing chronic dry eye symptoms or season allergies.
How Can Eye Drops Cause Blindness?
To the typical consumer, a contaminated eye drop bottle may not appear to be anything. But the reality is darker. Non-sterile solutions can be a portal of entry for bacteria, fungi, or even parasites, all of which can wreak havoc on the internal environment of the eye.
In 2023, when contaminated eye drops containing a drug-resistant bacteria strain caused 50 infections in 11 U.S. states. That outbreak caused one death and several cases of permanent blindness, highlighting just how perilous contaminated eye drops are.
More disturbing is the likelihood that consumers will unknowingly use these recalled products, particularly at the height of allergy season, when dry eyes are a frequent complaint and over-the-counter relief is in high demand.
If you have recently used one of the recalled eye drops, beware of these warning signs of potential infection:
- Redness or swelling of the eye
- Pain or stinging
- Blurred vision or visual changes
- Abnormal discharge or watery eyes
- Increased sensitivity to light
Although no health problems have yet been officially reported regarding this particular recall, an early intervention is always the best course of action. If infection is suspected, stop using at once and seek an ophthalmologist. AvKARE has put out a strong notice:
Immediately stop using the recalled products. Retailers are requested to pull the products from store shelves and return all unsold merchandise to the distributor for a full refund, including the cost of return shipping.
The recall was initiated by BRS Analytical Service, LLC, an independent laboratory that tests pharmaceuticals for compliance. The action demonstrates growing scrutiny from the FDA, particularly after last year's death from eye drops.
Although the identity of the manufacturer is not revealed, the FDA has not eliminated further action, particularly if more products prove to be dangerous.
What This Means For Your Eye Health?
This event brings to the fore the larger issues regarding the regulation and testing of over-the-counter (OTC) eye care products. It acts as a reminder for consumers and medical professionals alike to ensure that proper eye hygiene, product handling, and consultation with medical practitioners at the right time are ensured.
Even synthetic tears, deemed by most to be harmless, may induce temporary blurring of the vision, allergic reactions, or introduce irritants in the form of preservatives or thickeners if improperly used. No prescription or OTC eye drop should be employed for longer than recommended, and sharing usage is strictly avoided.
The patient should also understand that improper packaging and expired usage can weaken the product and even inflict more harm. Always examine the seal and expiration date and never use bottles with broken or tampered packages.
What To Do If Your Eyes Are Affected?
You may see a complete list of the products recalled and lot numbers on the FDA or AvKARE's official notice of recall webpage. If you already have the affected eye drops:
- Discontinue use immediately.
- Place them in a sealed bag to avoid accidental opening.
- Get a refund or disposal guidance from your retailer.
- Consult a healthcare professional if you're experiencing symptoms.
Medical practitioners are also cautioned to inform the FDA's MedWatch Safety Information and Adverse Event Reporting Program about any product quality issues or adverse events.
Stay up to date, read labels attentively, and never settle for the safety of what you put in your eyes. If you use eye drops frequently, think about talking to a certified ophthalmologist to discuss preservative-free options and individualized choices that are safer for long-term use.
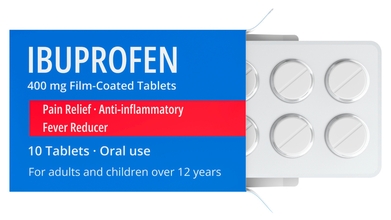
Metal Particle Found In Ibuprofen, Drug Now Recalled
Credits: iStock
Ibuflam tablets by pharmaceutical manufacturer Zentiva under the brand name Lichtenstein has been recalled. The 600mg tablets of ibuprofen products were recalled due to contamination. As per several media reports, a piece of metal was found in the tablet which triggered the recall. As per the manufacturer, the affected tablet are the 50-tablet packs with the batch number 5R02514.
Zentiva stated that the recall is explicitly directed at wholesalers and pharmacies, as the company confirmed upon request and the recall is not directed on patients. This measure was taken as a precautionary measure and in consultation with the relevant regulatory authorities.
As of now, patients have the option to return packages from the affected batch and receiving a replacement.
How Can You Return And Receive A New Ibuprofen Prescription?
As per Zentiva, the standard procedure of returning packages from the affected batch and receiving a replacement could be availed. For instance, if the package is almost empty, the standard procedure does not need to be followed, as this could potentially be considered a new prescription. However, patients will have the option of returning packages with the batch number.
A Zentiva spokesperson explained, "The possibility of obtaining a replacement through the supplying pharmacy in case of suspected quality defects always exists. Any connection to the recall lies solely in the fact that the suspicion regarding this batch can generally be considered justified."
This is not a case of re-dispensing under the replacement procedure. It is important to note that when a replacement pack is dispensed in response to a quality complaint, the affected medicinal product is not being placed on the market again, but rather a previously prescribed and supposedly defective medicinal product is being replaced.
Read: THIS Alternative Could Help Prevent Gut Damage Caused By Painkillers
What Is Ibuflam Or Ibuprofen?
Ibuprofen is a nonsteroidal anti-inflammatory drug or NSAID that is used to relieve pain, fever, and inflammation. Ibuflam is a brand name which is a combination medication containing ibuprofen and paracetamol or acetaminophen, which is also designed for enhanced pain relief.
As per the World Health Organization (WHO), ibuprofen is on the list of essential medication. The medicine is used by mouth as a syrup or tablet, and could also be applied directly to the skin as a mousse, gel, or spray.
The uses include:
- fever
- inflammation
- headache
- menstrual pain
- common cold
- toothache
- back pain
- arthritis
- sprains
The medicine could have some side effects, which could include pain, diarrhea or constipation, nausea and vomiting, dyspepsia, bloating, dizziness, headache, nervousness, skin rash, tinnitus, edema or fluid retention.
Who Should Not Use Ibuprofen?
Anyone with:
- repeated stomach problems like heartburn
- stomach ulcers
- bleeding problems
- high blood pressure
- heart disease
- kidney disease
- age over 60 years
OR
Anyone who:
- has taken a diuretic
- is using other NSAIDs or pain relief medication
- is using anticoagulants
- is receiving treatment for any serious condition
- has chickenpox or shingles
- has Crohn's disease or ulcerative colitis
- has liver problems
Leucovorin Approved By FDA But For A Rare Genetic Disease, Not For Autism
Credits: Reuters
The US Food and Drug Administration approved leucovorin, but not for what White House had recommended for in its September briefing. FDA has approved the prescription drug leucovorin for cerebral folate deficiency in the receptor 1 gene, a genetic condition that is estimated to affect only about 1 in 1 million people.
In September, the White House touted leucovorin as a potential treatment for children with autism. Health and Me previously reported on how parents were struggling to find the prescription drug, while not enough scientific evidence was there to support the claim.
What Is Leucovorin?
It is a high-dose B vitamin, which is commonly used as a treatment to counteract the side effects of chemotherapy. It has been approved by the FDA for cerebral folate deficiency in the receptor 1 gene. The disease is rare and fewer than 50 cases have been identified worldwide. FDA Commissioner Dr Marty Makary called this approval "a significant milestone" for patients with the condition.
However, in September last year, Markary and US Health and Human Services Secretary Robert F Kennedy Jr said that the drug could help children with autism. Kennedy, standing next to US President Donald Trump, said, they had "identified an exciting therapy that may benefit large number of children who suffer from autism."
CNN reported that in Austin, Texas, Meagan Johnson, a mother with an autistic son of three years of age, spent four days calling pharmacies across the region searching for leucovorin. She contacted nearly 40 pharmacies around her home in Pflugerville, hoping to locate the medication. Health and Me reported how getting the prescription turned out to be far harder than obtaining it.
This update on leucovorin, notes CNN have hit some families hard, especially the families of children with autism. This is because of the September briefing and when Trump also said that painkiller Tylenol could in fact cause autism. The drug leucovorin was used as something that could give them hope, however, with the update in its label, parents efforts to find the prescription drugs could feel like in vain.
The Rare Condition For Which Drug Got Its Approval
The drug got its approval for folate deficiency in the receptor 1 gene. This is a defect that causes proteins that guide folate into the brain to malfunction. As a result, normal levels of folate is found in the blood, but not in the brain or nervous system, due to which tasks like thinking, speech and movement could not be performed.
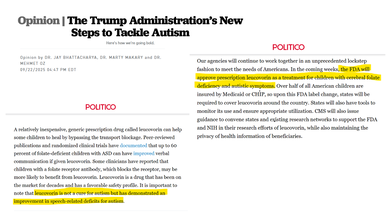
In an op/ed published in Politico and reposted on the White House website last fall, Markary said that his agency would "approve prescription leucovorin as a treatment for children with cerebral folate deficiency and autistic symptoms". However, he did not that it cause not a cure for autism, but there have been studies that show that it could help children with folate deficiency and autism to improve speech by roughly 60 per cent. The Trump administration made a bold move to expand the usage of the drug by "opening the door to the first FDA-recognized treatment for autism." wrote Markary and other HHS leaders.
AIIMS And ISRO Sign MoU To Advance Research In Space Medicine

Credits: Dr P Sarat Chandra (LinkedIn)
All India Institute of Medical Sciences (AIIMS) and the Indian Space Research Organisation (ISRO) signed a Memorandum of Understanding (MoU) on Monday to collaborate on advancing research in space medicine.
The MoU was signed between M Srinivas, Director (AIIMS), New Delhi and Dinesh Kumar Singh, Director, Human Space Flight Centre (HSFC), ISRO. The event brought together senior leadership from AIIMS and ISRO, including ISRO Chairman V Narayanan and Secretary, Department of Space, as well as Deans, Heads of Departments, faculty members, and studies from AIIMS New Delhi.
Also Read: Is There A Link Between Your Kidney Health And Other Chronic Diseases? Study Says Yes
As per the news agency Asian News International (ANI), a press release of the collaboration noted for a cooperative framework for ground-based and space-based research in Space Medicine. The press release also noted that the research will cover areas like human physiology, cardiovascular and autonomic regulation, musculoskeletal health in microgravity, microbiome and immunology, genomics and biomarkers and behavioral health.
“This MoU will give us the escape velocity to venture together into the field of space medicine. Collaborative research between AIIMS and ISRO will benefit patients, the nation, and ultimately humankind. As India moves toward Viksit Bharat by 2047, we hope to emerge as a Vishwaguru in space medicine as well,” said Prof M Srinivas, Director, AIIMS, New Delhi.

Chairman of ISRO Dr V Narayanan and Secretary, Department of Space also highlighted India’s remarkable journey in space programme. He noted how India evolved from the time when rockets and equipment were transported on bicycles and bullock carts to today, where it is going to be leading medical and research institutions like AIIMS to strengthen India’s human spaceflight capabilities.
The programme also featured a presentation on AIIMS New Delhi’s ongoing work in space medicine research, delivered by Prof. KK Deepak, former Head of the Department of Physiology. The event was attended by Heads of Departments and faculty members from across AIIMS New Delhi, along with representatives from the Resident Doctors Association (RDA), the AIIMS Student Association (ASA), and the Society of Young Scientists (SYS).
The collaboration represents an important step toward enhancing India’s capabilities in human spaceflight and biomedical research. It also aligns with the country’s broader scientific ambitions as India moves toward its centenary in 2047 and the vision of Viksit Bharat.
What Do We Already Know About Space Medicine?
As per a 2017 study in the British Journal of Anaesthesia, space medicine is fundamental to the human exploration of space. It supports survival, function and performance in this challenging as potentially lethal environment. It is international, intercultural and interdisciplinary, operating at the boundaries of exploration, science, technology and medicine.
Space medicine specialists play a crucial role in safeguarding astronauts and other spaceflight participants. Known as “flight surgeons,” they help develop strategies to protect the health, safety, and performance of space travellers in the extreme conditions of space. Their responsibilities span every stage of a mission — from selection and training to in-flight care, post-mission rehabilitation, and monitoring long-term health effects.
With inputs from ANI
© 2024 Bennett, Coleman & Company Limited

