- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
CDC Warns Travelers of Measles Risk at Airports and Popular Destinations

Credits: Canva
As the summer travel season peaks, the Centers for Disease Control and Prevention (CDC) has issued an important advisory to travelers about the re-emergence of measles in the US, specifically highlighting the risk of contracting the highly contagious disease at airports and other crowded transit points. Once being declared eradicated from the United States in 2000, measles has has made a unsettling comeback, fueled primarily by declining vaccination rates and global travel. With more than 1,100 reported cases across the country this year alone, the CDC's warning is a harsh reminder that this preventable illness is a very real and present danger—particularly in areas thick with travelers.
For generations, measles was a thing of the past in the U.S. due to widespread vaccination campaigns. The disease was almost eradicated over the last two decades, with only sporadic imported cases and small outbreaks occurring. Recent trends, though, have turned this tide of progress around. This year's figure of 1,157 cases already exceeds numbers from earlier years, indicating a public health problem not witnessed since 2019, when a large-scale outbreak rocked communities.
Much of this increase stems from a multistate outbreak concentrated in West Texas, but the risk is far from localized. The CDC highlights that the virus is highly contagious and can spread rapidly in places like airports, train stations, planes, crowded events, and tourist attractions—anywhere large numbers of people converge, particularly if some are unvaccinated or susceptible.
Why Airports and Travel Hubs Are High-Risk Zones?
Airports stand out for their ability to transmit disease. They are gateways to and from around the world with tightly packed crowds, frequently crowded waiting rooms, and intimate mingling among travelers from all parts of the globe—some from nations where measles remains endemic. The CDC travel notice emphasizes that travelers can catch measles while in these locations, then return home with the virus and spread it further.
The extreme infectiousness of measles makes it so that casual exposure to an infected individual or contact with contaminated surfaces can result in infection. Fever, cough, rash, and serious complications such as seizures and blindness are all symptoms that make it a dangerous virus, particularly for infants, pregnant women, and the immunocompromised.
Why Is Measles Surging?
The resurgence is largely due to vaccine hesitancy and disinformation that have caused some parents to skip or postpone childhood vaccinations. The implications are evident: areas of under-immunized communities are fertile breeding grounds for measles outbreaks. Anti-vaccine attitudes, in some cases spurred on by misinformation and suspicion of medical institutions, have been a prime force behind the current resurgence.
Adding to this problem is the amount of travel anticipated this summer. As millions of American travelers flood airports, trains, and vacation spots, the risk of transmission increases, particularly in areas with lower vaccination levels.
Vaccine Guidance and Precautions for Travelers
The CDC advises all international travelers to make sure they are up to date with their measles vaccination, including two doses of the measles-mumps-rubella (MMR) vaccine at least 28 days apart. Infants 6 to 11 months old traveling internationally should also receive an additional dose of MMR vaccine prior to travel.
Those born prior to 1957 are presumed to have had natural immunity from childhood exposure and thus are not recommended to be vaccinated by the CDC or Texas public health officials except in the case of healthcare personnel who work in outbreak areas, for whom two doses should be administered regardless of year of birth.
Some individuals, like pregnant women, infants younger than six months, and individuals with severely compromised immune systems, are not eligible for the vaccine. These persons should speak with their physicians regarding their individual risk and possible preventive action, like immunoglobulin therapy, that will provide temporary protection once exposed.
How to Stay Safe While Traveling?
Experts recommend that travelers take a proactive role by getting their immunity status checked sufficiently in advance of their travel—ideally six weeks or more ahead. This can include checking vaccination history, laboratory tests, or evidence of previous infection. Suspected exposure to measles calls for immediate medical consultation, since early administration of MMR vaccine or immunoglobulin can cut down on disease severity.
Along with vaccination, routine preventive precautions are important: frequent washing of hands, avoidance of contact with the sick, remaining in well-ventilated places, and use of masks when required, particularly for those with high risks of developing complications.
Travelers need to prepare as well by bringing along medications that are necessary and learning about healthcare facilities in the destinations. Checking the CDC's travel health notices may offer current information on risks and vaccine requirements for countries.
The recent measles outbreak is a cautionary example of the vulnerability of disease elimination efforts. Even as vaccination campaigns have long kept measles at bay, recent slippages risk undoing decades of gains. The warnings from the CDC occur at a time when wider concerns exist regarding vaccine misinformation and waning public trust in immunizations.
The consequences are dire. Measles is not a harmless childhood disease; it can result in severe complications, hospitalization, and even death. Already this year, at least two children and one adult have died from measles-related complications in the United States, and these figures may increase as travel continues and outbreaks multiply.
Travelers need to understand that immunity is the best defense against measles. Howsoever healthy your living style, germs do not distinguish between people—germs infect the unimmunized, the weak, and those who come in close proximity to carriers equally.
‘Contaminated’ wet wipes linked to fatal bacterial infections, death in UK

Credit: Canva
The UK Health Security Agency (HSA) and the Medicines and Healthcare products Regulatory Agency (MHRA) have warned people against using non-sterile alcohol-free wipes in their homes and first aid kits, over their link to a fatal bacterial infection, which has also led to death in the country.
After testing almost 200 products, the officials identified Burkholderia stabilis -- a type of bacteria found naturally in the environment, including in soil and water -- in four brands of skin cleansing wipes intended for first aid.
These brands of non-sterile alcohol-free wipes have been contaminated and have caused serious infection with the B. stabilis. These include:
- ValueAid Alcohol Free Cleansing Wipes
- Microsafe Moist Wipe Alcohol Free
- Steroplast Sterowipe Alcohol Free Cleansing Wipes
- Reliwipe Alcohol Free Cleansing Wipes
While B. stabilis rarely causes infection in healthy individuals, it can be dangerous to people with a weakened immune system, cystic fibrosis, or malignancy, the authorities said. Further, patients using intravenous lines at home are also at higher risk of developing infection.
The MHRA had, in 2025, also issued a warning against the four products.
“There have been 59 confirmed cases of Burkholderia stabilis associated with some non-sterile alcohol-free wipe products -- identified in an outbreak in the United Kingdom from January 2018 to 3 February 2026,” the joint statement said.
“A small number of cases continue to be detected. These have included some serious infections which have required hospital treatment, and one death has been attributed to Burkholderia stabilis infection,” it added.
The health agencies also stressed that only wipes marked as “sterile” should be used on broken skin and only used to clean intravenous lines if instructed by a patient’s medical team.
“We are reminding the public not to use, and to dispose of, certain non-sterile alcohol-free wipes, which have been linked to an outbreak of Burkholderia stabilis,” said James Elston, consultant in epidemiology and public health at UKHSA.
“Those who still have any of the affected products should stop using them immediately and dispose of them in standard household waste,” the UKHSA added.
A recent report by Eurosurveillance, early this month, also confirmed 59 cases of B. stabilis associated with contaminated non-sterile alcohol-free wipe products in the UK as of February.
Also read: What Kind Of Wipes Should You Use To Clean Wounds? Here's What UKHSA Suggests
Key Symptoms To Watch For
Anyone who has used non-sterile, alcohol-free wipes does not need to seek medical care unless they develop symptoms of infection, such as:
Wound infection: redness, swelling, warmth, increased pain, or pus/drainage from the site
Infection involving an IV line: redness, swelling, or pain at the insertion site, along with fever or chills
Safety Advice by UKHSA And MHRA
The UKHSA offers the following guidance for safe wipe use:
- Use only sterile wipes on broken skin, wounds, or scratches to reduce the risk of germs entering the body.
- Check that your first aid kit contains only wipes labelled “sterile,” ensure the packaging is intact, and dispose of any out-of-date or damaged products.
- Only use wipes on IV lines if specifically instructed by your healthcare team.
Dr Alison Cave, MHRA Chief Safety Officer, emphasized that wipes intended for medical purposes are classed as medicines.
These products do not have the necessary medicines authorization, and steps are being taken to enforce compliance.
“If you have these wipes at home or in a first aid kit, check the label and only use wipes marked as ‘sterile’ on broken skin,” Dr. Cave advised.
“Healthcare professionals should follow instructions provided in the national patient safety alert.”
Anyone who has used alcohol-free wipes and is concerned about possible infection should speak to a healthcare professional.
Passive Euthanasia: Harish Rana’s Case May Reshape End-of-life Protocols, Say Experts
Credit: iStock
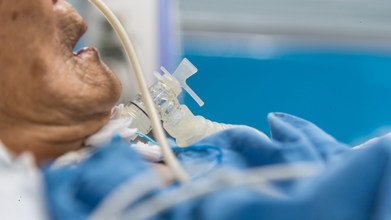
The Supreme Court of India, in a landmark decision, authorized the removal of life support for Harish Rana, a 31-year-old man in a vegetative state since 2013.
This marks the country's first Court-approved case of passive euthanasia without a prior living will. The Court ruled that the "right to die with dignity" is a fundamental part of the right to life under Article 21.
Also read: Supreme Court Allows 1st Passive Euthanasia For Man In Vegetative State For 13 Years
Speaking to HealthandMe, the experts said that the landmark ruling will enable families and doctors to make compassionate decisions and may also influence end-of-life protocols.
There are several medical conditions where patients undergo prolonged suffering despite treatment, with no realistic scope for recovery, sometimes for decades.
“This judgment could have a significant impact on end-of-life care practices in Indian ICUs. Many patients remain in prolonged vegetative states with no meaningful quality of life, often sustained only through artificial life support,” Dr. Sandeep Dewan, Senior Director, Critical Care & Chairman ECMO Program, Fortis Gurugram, told this publication.
“The ruling reinforces that while preserving life is important, the quality and dignity of life must also be considered, and it provides clearer pathways for families and doctors to make compassionate decisions in such situations,” he added.
Harish was a BTech student in Chandigarh who suffered severe traumatic brain injury after falling from the fourth floor of his paying guest accommodation in August 2013.
Since then, he has remained bedridden and was being treated with Clinically Administered Nutrition (CAN), where surgically installed PEG tubes helped him with breathing and nutrition.
The apex Court, in its ruling, noted that it can just prolong his biological existence, but it will not lead to any therapeutic improvement.
With the Harish Rana judgment, the apex Court today clarified how passive euthanasia should be applied in cases where a patient’s life is being supported by feeding tubes.
The top Court also waived off the reconsideration period of 30 days and noted that the medical treatment, including the CAN administered to the patient, can be withdrawn or withheld.
"Doctors and hospitals have often been reluctant to stop tube feeding in such patients, fearing that it could be interpreted as 'starving the patient to death',” Dr. Rajeev Jayadevan, Ex-President of IMA Cochin and Convener of the Research Cell, Kerala, told HealthandMe.
“Today’s ruling clarifies that artificial nutrition and hydration are indeed forms of medical treatment. Therefore, withholding such artificial feeding can be considered withdrawal of life-sustaining medical support in situations where treatment offers no prospect of recovery and only prolongs suffering,” he added.
Passive Euthanasia In India
Passive Euthanasia allows a terminally ill or irreversibly comatose patient to die naturally. It involves deliberately withholding or withdrawing life-sustaining treatments (like ventilators, feeding tubes, or medication). It has been legal since 2018, but under strict guidelines.
On the other hand, active euthanasia or assisted suicide for terminally ill patients is legal in several countries, but is not permitted in India.
The Aruna Shanbaug Case (2011) paved the way for passive euthanasia in India.
Shanbaug was a nurse at Mumbai's KEM hospital who remained in a vegetative state for 42 years after an assault in 1973. The hospital staff cared for her and did not stop treatment till she passed away naturally in 2015.
However, in the 2011 Aruna Shanbaug judgment, the SC allowed passive euthanasia by permitting the withdrawal or withholding of life-sustaining treatment under strict legal safeguards.
This framework was further clarified in the 2018 Common Cause judgment, which recognized advance directives or living wills.
Later in 2023, the SC modified the guidelines, noting that withdrawal of life support is permissible only after the approval of the Primary and Secondary Medical Boards.
A Living Will
Dr. Jayadevan noted that, as death is a certainty for all who are living, greater awareness must be created on adults preparing a "Living Will or Advanced Directive".
A Living Will is essentially made when individuals are "still in good health— documenting one’s preference for specific treatment measures in the event of a terminal illness occurring in the future”.
“This will help relatives and doctors to take the right decisions and avoid unnecessary treatment measures in such situations. Unlike the conventional Will that is executed after death, a Living Will is implemented when a person is still alive,” the doctor said.
Supreme Court Allows 1st Passive Euthanasia For Man In Vegetative State For 13 Years

Credit: iStock
In a landmark judgement, the Supreme Court today allowed passive euthanasia for a 32-year-old man, living in a vegetative state for the last 13 years.
A bench comprising Justice JB Pardiwala and Justice KV Viswanathan allowed the withdrawal of life support for Harish Rana, a resident of Ghaziabad, who has been in a coma and kept alive on tubes for breathing and nutrition after sustaining severe head injuries following a fall from a building in 2013 in Chandigarh.
It is the first known case of a court-ordered passive euthanasia in India, since it was legalised in 2018 and modified in 2023, recognizing the fundamental right to die with dignity.
"Harish Rana, presently aged 32 years, was once a young, bright boy. He met with a tragic life-altering accident after a fall from the fourth floor of his paying guest accommodation. His brain injury left him in a condition of Persistent Vegetative State (PSV) with 100 percent quadraplegia... Medical reports show that his medical condition has not improved in the past 13 years," LiveLaw quoted the bench as saying.
The Court noted that the continuation of his treatment -- Clinically Administered Nutrition (CAN) via surgically installed PEG tubes -- can just prolong his biological existence but will not lead to any therapeutic improvement.
What Is The Case Of Harish Rana?
Harish was a BTech student in Chandigarh who suffered severe traumatic brain injury after falling from the fourth floor of his paying guest accommodation in August 2013.
Since then, he has remained bedridden and dependent on others for all activities of daily life.
Harish's father, the petitioner, first approached the Delhi High Court in 2024, seeking permission for passive euthanasia, but was rejected as the patient was not terminally ill.
The same year, the petitioner knocked on the doors of the Supreme Court, which, though it refused to entertain the plea, directed the Uttar Pradesh government to bear the treatment expenses.
In 2025, the petitioner filed a miscellaneous application in the Supreme Court, noting that Harish's condition had no scope for improvement.
The Court then directed the constitution of a Primary Medical Board led by the District Hospital in Noida to examine his health, as well as a Secondary Medical Board constituted by the All India Institute of Medical Sciences (AIIMS).
After perusing the report, Justice Pardiwala remarked that it's a "sad report" and the man can't continue to live like this. Before passing the final order, the Court met the parents, LiveLaw reported.
The Court has asked AIIMS to provide palliative care, so that the withdrawal of CAN can be given effect to.
To maintain the dignity of death, the apex Court said that the life support must be withdrawn with a tailored plan.
1st Passive Euthanasia: What's New From The 2018 Judgment
In 2018, a five-judge Constitution Bench had recognized and given sanction for passive euthanasia, and living will/advance directives.
Later in 2023, the SC modified the guidelines, noting that withdrawal of life support is permissible only after the approval of the Primary and Secondary Medical Boards.
With the Harish Rana judgment, the apex Court today clarified how passive euthanasia should be applied in cases where a patient’s life is being supported by feeding tubes.
The top Court waived off the reconsideration period of 30 days and noted that the medical treatment, including the CAN administered to the patient, can be withdrawn or withheld.
What is Passive Euthanasia
Passive Euthanasia allows a terminally ill or irreversibly comatose patient to die naturally. It involves deliberately withholding or withdrawing life-sustaining treatments (like ventilators, feeding tubes, or medication). It has been legal since 2018, but under strict guidelines.
In Active Euthanasia, patients are administered a lethal injection to cause death. It is illegal in India and considered an offence.
The Aruna Shanbaug case in 2011 opened the door for passive euthanasia in India for the first time.
The top Court rejected euthanasia in the case of Shanbaug, a nurse at Mumbai's KEM hospital who was in a vegetative state for 42 years after an assault in 1973, as the hospital staff who cared for her for decades did not support stopping treatment.
Shanbaug continued to be under care and passed away naturally in 2015
However, in her case, the court made the judgment allowing for passive euthanasia in certain rare situations under strict conditions.
© 2024 Bennett, Coleman & Company Limited

