- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Delhi Isn't Just Breathing Toxic Air, But Also A Superbug That Resists Antibiotics

Credits: iStock
Delhi is not just breathing toxic air, but also a superbug through its air, which was found in a latest research conducted by the researchers are Jawaharlal Nehru University, published in Nature - Scientific Reports. Apart from the heavy particulate matter or the PM2.5, which are small enough to penetrate through one's bloodstream and cause blockage, Delhi's air also contains airborne bacteria, including staphylococci. The bacteria levels exceed the safety limit by 16-fold as provided by the World Health Organization (WHO).
According to scientists most of these bacteria are methicillin resistant and unaffected by multiple antibiotic classes. This has raised concerns about the hard to treat and life threatening air that people are breathing in Delhi, also responsible for causing respiratory infections. While Delhi's AQI may have improved in the past few days and come down to 'very poor' from 'severe', high levels of particulate matter still put many people at the risk of aggravated asthma and cardiovascular disease. However, the added finding of the antimicrobial resistant superbug also adds a new layer of threat to residents.
Also Read: Flu Cases On The Surge, ER Doctor Shares 5 Must-know Things About The Illness
Many have left the city already due to the toxic air. Aflah Ul Haque, a 33-year-old Media and Communications PR consultant had been living in Delhi since 2017. However, in 2024, he considered to move out. He is now living in Aligarh. While he did face difficulties to uproot himself from a metro city like Delhi, as it had become his comfort zone, it was a decision he had to take for a healthy life. Health and Me also previously reported stories of people who shifted or are actively considering shifting out of the city. Will more people consider the same move now that a superbug is found in the air? This is yet to be seen.
What Did PM Modi Say About Antimicrobial Resistance?
India is already fighting a battle with antimicrobial resistance or AMR, with the breakthrough of Zaynich, a drug to treat AMR, there is hope, however, one still needs to be extra careful. On December 28, 2025, Prime Minister Narendra Modi on the 129th edition of "Mann Ki Baat", spoke about AMR. He pointed out that this is a matter of concern and cited data from the Indian Council of Medical Research (ICMR) showing that antibiotics are becoming less effective against pneumonia and urinary tract infections. He also pointed out that in India, this is a crisis due to "thoughtless and indiscriminate use of antibiotics by people".
How Was The Study That Found Superbug In Delhi's Air Conducted?
In order to understand what people might be breathing in, the researchers collected air samples from a range of everyday locations across Delhi, such as:
- Crowded market areas
- Urban slum neighborhoods
- Residential apartments
- A sewage treatment plant located within the JNU campus
Both indoor and outdoor air samples were taken during summer, monsoon and winter to track how bacterial levels and resistance patterns shifted with the seasons.
Across every setting, airborne bacteria levels were far above the World Health Organization’s recommended safe limit for microbial exposure. In some winter samples, bacterial counts exceeded 16,000 colony-forming units per cubic meter, which is more than 16 times higher than the WHO guideline.
What raised even greater concern was the type of bacteria detected. Many belonged to the Staphylococcus group, which includes strains linked to skin infections, pneumonia, bloodstream infections and illnesses commonly acquired in hospitals.
According to the researchers, a large share of the airborne bacteria were identified as methicillin-resistant staphylococci (MRS), meaning they do not respond to commonly prescribed antibiotics. Among these:
73 per cent were multidrug-resistant, showing resistance to several classes of antibiotics
Many were resistant to macrolides, beta-lactams, trimethoprim, gentamicin and other commonly used drugs
Genetic analysis confirmed the presence of antibiotic resistance genes (ARGs)
Notably, 14 out of 36 multidrug-resistant samples carried the mecA gene, a well-known marker that makes bacteria resistant to methicillin and related antibiotics.
Why Is Winter Smog Linked To Drug-Resistant Bacteria?
The study showed that levels of airborne drug-resistant bacteria were highest in winter, while monsoon rains led to a sharp drop in outdoor contamination.
Researchers point to a few likely reasons:
- Poor air movement in winter traps pollutants and microbes near ground level
- High levels of particulate matter may give bacteria surfaces to cling to and spread
- Cooler temperatures and lower humidity allow bacteria to survive longer in the air
On the other hand, rainfall during the monsoon appears to clear bioaerosols from the air, reducing bacterial concentrations outdoors.
Metal Particle Found In Ibuprofen, Drug Now Recalled
Credits: iStock
Ibuflam tablets by pharmaceutical manufacturer Zentiva under the brand name Lichtenstein has been recalled. The 600mg tablets of ibuprofen products were recalled due to contamination. As per several media reports, a piece of metal was found in the tablet which triggered the recall. As per the manufacturer, the affected tablet are the 50-tablet packs with the batch number 5R02514.
Zentiva stated that the recall is explicitly directed at wholesalers and pharmacies, as the company confirmed upon request and the recall is not directed on patients. This measure was taken as a precautionary measure and in consultation with the relevant regulatory authorities.
As of now, patients have the option to return packages from the affected batch and receiving a replacement.
How Can You Return And Receive A New Ibuprofen Prescription?
As per Zentiva, the standard procedure of returning packages from the affected batch and receiving a replacement could be availed. For instance, if the package is almost empty, the standard procedure does not need to be followed, as this could potentially be considered a new prescription. However, patients will have the option of returning packages with the batch number.
A Zentiva spokesperson explained, "The possibility of obtaining a replacement through the supplying pharmacy in case of suspected quality defects always exists. Any connection to the recall lies solely in the fact that the suspicion regarding this batch can generally be considered justified."
This is not a case of re-dispensing under the replacement procedure. It is important to note that when a replacement pack is dispensed in response to a quality complaint, the affected medicinal product is not being placed on the market again, but rather a previously prescribed and supposedly defective medicinal product is being replaced.
Read: THIS Alternative Could Help Prevent Gut Damage Caused By Painkillers
What Is Ibuflam Or Ibuprofen?
Ibuprofen is a nonsteroidal anti-inflammatory drug or NSAID that is used to relieve pain, fever, and inflammation. Ibuflam is a brand name which is a combination medication containing ibuprofen and paracetamol or acetaminophen, which is also designed for enhanced pain relief.
As per the World Health Organization (WHO), ibuprofen is on the list of essential medication. The medicine is used by mouth as a syrup or tablet, and could also be applied directly to the skin as a mousse, gel, or spray.
The uses include:
- fever
- inflammation
- headache
- menstrual pain
- common cold
- toothache
- back pain
- arthritis
- sprains
The medicine could have some side effects, which could include pain, diarrhea or constipation, nausea and vomiting, dyspepsia, bloating, dizziness, headache, nervousness, skin rash, tinnitus, edema or fluid retention.
Who Should Not Use Ibuprofen?
Anyone with:
- repeated stomach problems like heartburn
- stomach ulcers
- bleeding problems
- high blood pressure
- heart disease
- kidney disease
- age over 60 years
OR
Anyone who:
- has taken a diuretic
- is using other NSAIDs or pain relief medication
- is using anticoagulants
- is receiving treatment for any serious condition
- has chickenpox or shingles
- has Crohn's disease or ulcerative colitis
- has liver problems
Leucovorin Approved By FDA But For A Rare Genetic Disease, Not For Autism
Credits: Reuters
The US Food and Drug Administration approved leucovorin, but not for what White House had recommended for in its September briefing. FDA has approved the prescription drug leucovorin for cerebral folate deficiency in the receptor 1 gene, a genetic condition that is estimated to affect only about 1 in 1 million people.
In September, the White House touted leucovorin as a potential treatment for children with autism. Health and Me previously reported on how parents were struggling to find the prescription drug, while not enough scientific evidence was there to support the claim.
What Is Leucovorin?
It is a high-dose B vitamin, which is commonly used as a treatment to counteract the side effects of chemotherapy. It has been approved by the FDA for cerebral folate deficiency in the receptor 1 gene. The disease is rare and fewer than 50 cases have been identified worldwide. FDA Commissioner Dr Marty Makary called this approval "a significant milestone" for patients with the condition.
However, in September last year, Markary and US Health and Human Services Secretary Robert F Kennedy Jr said that the drug could help children with autism. Kennedy, standing next to US President Donald Trump, said, they had "identified an exciting therapy that may benefit large number of children who suffer from autism."
CNN reported that in Austin, Texas, Meagan Johnson, a mother with an autistic son of three years of age, spent four days calling pharmacies across the region searching for leucovorin. She contacted nearly 40 pharmacies around her home in Pflugerville, hoping to locate the medication. Health and Me reported how getting the prescription turned out to be far harder than obtaining it.
This update on leucovorin, notes CNN have hit some families hard, especially the families of children with autism. This is because of the September briefing and when Trump also said that painkiller Tylenol could in fact cause autism. The drug leucovorin was used as something that could give them hope, however, with the update in its label, parents efforts to find the prescription drugs could feel like in vain.
The Rare Condition For Which Drug Got Its Approval
The drug got its approval for folate deficiency in the receptor 1 gene. This is a defect that causes proteins that guide folate into the brain to malfunction. As a result, normal levels of folate is found in the blood, but not in the brain or nervous system, due to which tasks like thinking, speech and movement could not be performed.
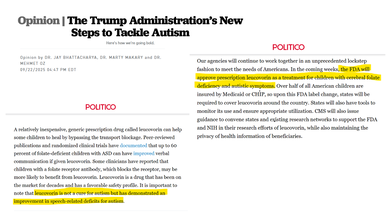
In an op/ed published in Politico and reposted on the White House website last fall, Markary said that his agency would "approve prescription leucovorin as a treatment for children with cerebral folate deficiency and autistic symptoms". However, he did not that it cause not a cure for autism, but there have been studies that show that it could help children with folate deficiency and autism to improve speech by roughly 60 per cent. The Trump administration made a bold move to expand the usage of the drug by "opening the door to the first FDA-recognized treatment for autism." wrote Markary and other HHS leaders.
AIIMS And ISRO Sign MoU To Advance Research In Space Medicine

Credits: Dr P Sarat Chandra (LinkedIn)
All India Institute of Medical Sciences (AIIMS) and the Indian Space Research Organisation (ISRO) signed a Memorandum of Understanding (MoU) on Monday to collaborate on advancing research in space medicine.
The MoU was signed between M Srinivas, Director (AIIMS), New Delhi and Dinesh Kumar Singh, Director, Human Space Flight Centre (HSFC), ISRO. The event brought together senior leadership from AIIMS and ISRO, including ISRO Chairman V Narayanan and Secretary, Department of Space, as well as Deans, Heads of Departments, faculty members, and studies from AIIMS New Delhi.
Also Read: Is There A Link Between Your Kidney Health And Other Chronic Diseases? Study Says Yes
As per the news agency Asian News International (ANI), a press release of the collaboration noted for a cooperative framework for ground-based and space-based research in Space Medicine. The press release also noted that the research will cover areas like human physiology, cardiovascular and autonomic regulation, musculoskeletal health in microgravity, microbiome and immunology, genomics and biomarkers and behavioral health.
“This MoU will give us the escape velocity to venture together into the field of space medicine. Collaborative research between AIIMS and ISRO will benefit patients, the nation, and ultimately humankind. As India moves toward Viksit Bharat by 2047, we hope to emerge as a Vishwaguru in space medicine as well,” said Prof M Srinivas, Director, AIIMS, New Delhi.

Chairman of ISRO Dr V Narayanan and Secretary, Department of Space also highlighted India’s remarkable journey in space programme. He noted how India evolved from the time when rockets and equipment were transported on bicycles and bullock carts to today, where it is going to be leading medical and research institutions like AIIMS to strengthen India’s human spaceflight capabilities.
The programme also featured a presentation on AIIMS New Delhi’s ongoing work in space medicine research, delivered by Prof. KK Deepak, former Head of the Department of Physiology. The event was attended by Heads of Departments and faculty members from across AIIMS New Delhi, along with representatives from the Resident Doctors Association (RDA), the AIIMS Student Association (ASA), and the Society of Young Scientists (SYS).
The collaboration represents an important step toward enhancing India’s capabilities in human spaceflight and biomedical research. It also aligns with the country’s broader scientific ambitions as India moves toward its centenary in 2047 and the vision of Viksit Bharat.
What Do We Already Know About Space Medicine?
As per a 2017 study in the British Journal of Anaesthesia, space medicine is fundamental to the human exploration of space. It supports survival, function and performance in this challenging as potentially lethal environment. It is international, intercultural and interdisciplinary, operating at the boundaries of exploration, science, technology and medicine.
Space medicine specialists play a crucial role in safeguarding astronauts and other spaceflight participants. Known as “flight surgeons,” they help develop strategies to protect the health, safety, and performance of space travellers in the extreme conditions of space. Their responsibilities span every stage of a mission — from selection and training to in-flight care, post-mission rehabilitation, and monitoring long-term health effects.
With inputs from ANI
© 2024 Bennett, Coleman & Company Limited

