- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
India’s First Indigenous Dengue Vaccine Coming Soon; What To Expect?
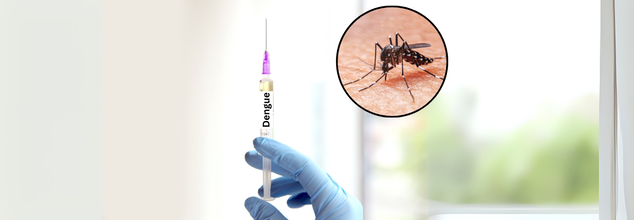
Dengue fever has been a public health crisis for decades, especially in tropical and subtropical regions, and India has been one of the worst-hit countries in recent years. With rising cases and fatal outbreaks, the country has required a reliable and inexpensive vaccine to prevent the spread of the disease. That changed with the launch of DengiAll, India's indigenous tetravalent dengue vaccine. After all these years of research, trials, and international collaboration, the vaccine is now in the last stage of testing, and a breakthrough seems at hand.
Developed by the Indian Council of Medical Research (ICMR) in collaboration with Panacea Biotech, DengiAll will be a game-changer for India's battle against dengue. The vaccine is currently in Phase 3 clinical trials, which are being conducted at 19 sites in 18 states and union territories with more than 10,335 healthy volunteers enrolled so far. The trials have shown promising results, with earlier phases (1 and 2) being successful for the vaccine to combat dengue across various strains.
The significance of the vaccine is not just its possibility in dengue prevention but also that it's an indigenous development, carefully crafted to fulfill India's needs. Balram Bhargava, former Director General of ICMR, who has played a key role in India's fight against COVID-19, ensured that India's first indigenous tetravalent dengue vaccine will be in the market by mid-2026—a landmark moment for India's health sector.
Dengue fever, transmitted by the Aedes aegypti mosquito, is one of the most common vector-borne diseases in India, especially prevalent during the monsoon season between May and September. The disease is characterized by fever, severe headache, joint pains, and sudden drop in platelet levels. It can, in severe cases, progress to dengue hemorrhagic fever (DHF) or dengue shock syndrome (DSS), which will prove fatal without urgent medical treatment. The World Health Organization (WHO) indicates that the worldwide incidence of dengue has grown eight times greater than two decades ago, thus emerging as a public health issue of major concern.
DengiAll is a tetravalent vaccine, and it has the goal of protecting against all four serotypes of the dengue virus (DENV-1, DENV-2, DENV-3, and DENV-4). This is a significant feature since dengue is a multi-serotype disease, and immunity to one serotype will not protect against others. In protecting against all four serotypes, the vaccine provides broad protection, which is extremely important in a country like India, where outbreaks are ever-present and in large part unpredictable.
The vaccine development is based on the TV003/TV005 strain, which was originally developed by the United States' National Institutes of Health (NIH). The strain was licensed to Panacea Biotech, a top Indian pharma company, and was adapted for domestic use. The company has achieved significant milestones in re-developing the vaccine to meet the specific needs of the Indian population, for whom it holds a process patent for development.
Highest Number of Cases Recorded In India
Approximately 188,401 cases were identified in 2017, with 325 deaths. Highest number of infections was reported from Tamil Nadu, followed by Kerala, Karnataka, then Punjab and West Bengal. In the 20th century, there was the significant rise in number of infections reported in 2003, 2010, 2012, and 2013, with an exponential increase beginning in 2015 onwards. During this time, repeated dengue outbreaks were reported in states of Andhra Pradesh, Goa, Delhi, Haryana, Karnataka, Gujarat, Kerala, Tamil Nadu, Punjab, and West Bengal. In 2017, dengue cases were to be the highest in India.
India, in particular, has experienced frequent outbreaks in West Bengal, Uttar Pradesh, Rajasthan, Bihar, Delhi, Kerala, and Tamil Nadu. The outbreaks have claimed thousands of lives and loaded the healthcare system with a great burden. Over 5 million cases of dengue have been reported to date as of 2023, with the Indian contribution playing a significant role in the surge. Since DengiAll is presently in its final stages of trials, the vaccine is set to reduce significantly the incidence of dengue and its complications in the country.
Why Dengue Cases Are On A Steady Rise?
Although India has been battling with dengue for years now, the illness has also been on the rise globally. The WHO states that the number of dengue cases reported globally has grown over twice in the past few years, with a total of 10.6 million cases having been reported thus far in the year 2024 alone. The growth is driven by various factors:
- Warm, humid temperatures offer ideal breeding grounds for mosquitoes.
- Overcrowded cities with poor waste management infrastructure facilitate the disease transmission.
- Increased international travel has led to dengue's expansion into non-endemic areas.
There is no cure for dengue known at the moment. It is treated with supportive care—hydration, management of fever, and close attention to platelet levels. If it is severe, hospitalization is required, and there are concerns regarding the burden this causes to the health system, especially in low-resource settings.
The presence of a dengue vaccine like DengiAll would be a welcome preventive. While a vaccine would not replace effective measures for mosquito control, it could be an extra tool in the public health arsenal, significantly reducing the number of severe cases and fatalities.
When Will Dengue Vaccine Hit the Market?
So far, DengiAll is in Phase 3 trials, and although it shows a great deal of promise, it should be kept in mind that the final approval phase could take time. If the ongoing trials continue to progress well, it is hoped that the vaccine would become available by mid-2026. This is a rough estimate, though, as it includes successful completion of the clinical trials and regulatory approvals.
Signs and Symptoms of Dengue Fever
Symptoms of dengue fever typically appear 4 to 10 days from the time of a bite from an infected mosquito and persist for 3 to 7 days. Asymptomatic cases are most common, but about 1 in 20 individuals will experience severe dengue when the initial symptoms start to resolve. Typical symptoms of dengue fever are:
- High fever
- Rash
- Severe pain behind the eyes
- Nausea and vomiting
- Muscle, bone, and joint pain
If you notice any of these symptoms, it is imperative that you receive medical care immediately, particularly if the symptoms are increasing.
How Does Vaccine Affects Dengue Treatment?
The DengiAll vaccine, India’s pioneering tetravalent dengue vaccine developed by Panacea Biotec, targets all four dengue serotypes, offering a preventive approach that could revolutionize dengue management. Dr. Divya Gopal, Consultant - Internal Medicine, Sir HN Reliance Foundation Hospital, Mumbai says, "Currently in Phase 3 trials with over 10,000 participants, it may reach markets by mid-2026 if successful. Unlike existing treatments, which focus on symptom management due to the absence of specific antivirals, DengiAll aims to reduce infection rates and disease severity, potentially lowering hospitalization and mortality, particularly during monsoon seasons. By shifting focus to prevention, DengiAll could ease the burden on healthcare systems and complement vector control efforts, significantly altering India’s dengue response strategy."
‘Contaminated’ wet wipes linked to fatal bacterial infections, death in UK

Credit: Canva
The UK Health Security Agency (HSA) and the Medicines and Healthcare products Regulatory Agency (MHRA) have warned people against using non-sterile alcohol-free wipes in their homes and first aid kits, over their link to a fatal bacterial infection, which has also led to death in the country.
After testing almost 200 products, the officials identified Burkholderia stabilis -- a type of bacteria found naturally in the environment, including in soil and water -- in four brands of skin cleansing wipes intended for first aid.
These brands of non-sterile alcohol-free wipes have been contaminated and have caused serious infection with the B. stabilis. These include:
- ValueAid Alcohol Free Cleansing Wipes
- Microsafe Moist Wipe Alcohol Free
- Steroplast Sterowipe Alcohol Free Cleansing Wipes
- Reliwipe Alcohol Free Cleansing Wipes
While B. stabilis rarely causes infection in healthy individuals, it can be dangerous to people with a weakened immune system, cystic fibrosis, or malignancy, the authorities said. Further, patients using intravenous lines at home are also at higher risk of developing infection.
The MHRA had, in 2025, also issued a warning against the four products.
“There have been 59 confirmed cases of Burkholderia stabilis associated with some non-sterile alcohol-free wipe products -- identified in an outbreak in the United Kingdom from January 2018 to 3 February 2026,” the joint statement said.
“A small number of cases continue to be detected. These have included some serious infections which have required hospital treatment, and one death has been attributed to Burkholderia stabilis infection,” it added.
The health agencies also stressed that only wipes marked as “sterile” should be used on broken skin and only used to clean intravenous lines if instructed by a patient’s medical team.
“We are reminding the public not to use, and to dispose of, certain non-sterile alcohol-free wipes, which have been linked to an outbreak of Burkholderia stabilis,” said James Elston, consultant in epidemiology and public health at UKHSA.
“Those who still have any of the affected products should stop using them immediately and dispose of them in standard household waste,” the UKHSA added.
A recent report by Eurosurveillance, early this month, also confirmed 59 cases of B. stabilis associated with contaminated non-sterile alcohol-free wipe products in the UK as of February.
Also read: What Kind Of Wipes Should You Use To Clean Wounds? Here's What UKHSA Suggests
Key Symptoms To Watch For
Anyone who has used non-sterile, alcohol-free wipes does not need to seek medical care unless they develop symptoms of infection, such as:
Wound infection: redness, swelling, warmth, increased pain, or pus/drainage from the site
Infection involving an IV line: redness, swelling, or pain at the insertion site, along with fever or chills
Safety Advice by UKHSA And MHRA
The UKHSA offers the following guidance for safe wipe use:
- Use only sterile wipes on broken skin, wounds, or scratches to reduce the risk of germs entering the body.
- Check that your first aid kit contains only wipes labelled “sterile,” ensure the packaging is intact, and dispose of any out-of-date or damaged products.
- Only use wipes on IV lines if specifically instructed by your healthcare team.
Dr Alison Cave, MHRA Chief Safety Officer, emphasized that wipes intended for medical purposes are classed as medicines.
These products do not have the necessary medicines authorization, and steps are being taken to enforce compliance.
“If you have these wipes at home or in a first aid kit, check the label and only use wipes marked as ‘sterile’ on broken skin,” Dr. Cave advised.
“Healthcare professionals should follow instructions provided in the national patient safety alert.”
Anyone who has used alcohol-free wipes and is concerned about possible infection should speak to a healthcare professional.
Passive Euthanasia: Harish Rana’s Case May Reshape End-of-life Protocols, Say Experts
Credit: iStock
The Supreme Court of India, in a landmark decision, authorized the removal of life support for Harish Rana, a 31-year-old man in a vegetative state since 2013.
This marks the country's first Court-approved case of passive euthanasia without a prior living will. The Court ruled that the "right to die with dignity" is a fundamental part of the right to life under Article 21.
Also read: Supreme Court Allows 1st Passive Euthanasia For Man In Vegetative State For 13 Years
Speaking to HealthandMe, the experts said that the landmark ruling will enable families and doctors to make compassionate decisions and may also influence end-of-life protocols.
There are several medical conditions where patients undergo prolonged suffering despite treatment, with no realistic scope for recovery, sometimes for decades.
“This judgment could have a significant impact on end-of-life care practices in Indian ICUs. Many patients remain in prolonged vegetative states with no meaningful quality of life, often sustained only through artificial life support,” Dr. Sandeep Dewan, Senior Director, Critical Care & Chairman ECMO Program, Fortis Gurugram, told this publication.
“The ruling reinforces that while preserving life is important, the quality and dignity of life must also be considered, and it provides clearer pathways for families and doctors to make compassionate decisions in such situations,” he added.
Harish was a BTech student in Chandigarh who suffered severe traumatic brain injury after falling from the fourth floor of his paying guest accommodation in August 2013.
Since then, he has remained bedridden and was being treated with Clinically Administered Nutrition (CAN), where surgically installed PEG tubes helped him with breathing and nutrition.
The apex Court, in its ruling, noted that it can just prolong his biological existence, but it will not lead to any therapeutic improvement.
With the Harish Rana judgment, the apex Court today clarified how passive euthanasia should be applied in cases where a patient’s life is being supported by feeding tubes.
The top Court also waived off the reconsideration period of 30 days and noted that the medical treatment, including the CAN administered to the patient, can be withdrawn or withheld.
"Doctors and hospitals have often been reluctant to stop tube feeding in such patients, fearing that it could be interpreted as 'starving the patient to death',” Dr. Rajeev Jayadevan, Ex-President of IMA Cochin and Convener of the Research Cell, Kerala, told HealthandMe.
“Today’s ruling clarifies that artificial nutrition and hydration are indeed forms of medical treatment. Therefore, withholding such artificial feeding can be considered withdrawal of life-sustaining medical support in situations where treatment offers no prospect of recovery and only prolongs suffering,” he added.
Passive Euthanasia In India
Passive Euthanasia allows a terminally ill or irreversibly comatose patient to die naturally. It involves deliberately withholding or withdrawing life-sustaining treatments (like ventilators, feeding tubes, or medication). It has been legal since 2018, but under strict guidelines.
On the other hand, active euthanasia or assisted suicide for terminally ill patients is legal in several countries, but is not permitted in India.
The Aruna Shanbaug Case (2011) paved the way for passive euthanasia in India.
Shanbaug was a nurse at Mumbai's KEM hospital who remained in a vegetative state for 42 years after an assault in 1973. The hospital staff cared for her and did not stop treatment till she passed away naturally in 2015.
However, in the 2011 Aruna Shanbaug judgment, the SC allowed passive euthanasia by permitting the withdrawal or withholding of life-sustaining treatment under strict legal safeguards.
This framework was further clarified in the 2018 Common Cause judgment, which recognized advance directives or living wills.
Later in 2023, the SC modified the guidelines, noting that withdrawal of life support is permissible only after the approval of the Primary and Secondary Medical Boards.
A Living Will
Dr. Jayadevan noted that, as death is a certainty for all who are living, greater awareness must be created on adults preparing a "Living Will or Advanced Directive".
A Living Will is essentially made when individuals are "still in good health— documenting one’s preference for specific treatment measures in the event of a terminal illness occurring in the future”.
“This will help relatives and doctors to take the right decisions and avoid unnecessary treatment measures in such situations. Unlike the conventional Will that is executed after death, a Living Will is implemented when a person is still alive,” the doctor said.
Supreme Court Allows 1st Passive Euthanasia For Man In Vegetative State For 13 Years

Credit: iStock
In a landmark judgement, the Supreme Court today allowed passive euthanasia for a 32-year-old man, living in a vegetative state for the last 13 years.
A bench comprising Justice JB Pardiwala and Justice KV Viswanathan allowed the withdrawal of life support for Harish Rana, a resident of Ghaziabad, who has been in a coma and kept alive on tubes for breathing and nutrition after sustaining severe head injuries following a fall from a building in 2013 in Chandigarh.
Also Read: Metal Particle Found In Ibuprofen, Drug Now Recalled
It is the first known case of a court-ordered passive euthanasia in India, since it was legalised in 2018 and modified in 2023, recognizing the fundamental right to die with dignity.
"Harish Rana, presently aged 32 years, was once a young, bright boy. He met with a tragic life-altering accident after a fall from the fourth floor of his paying guest accommodation. His brain injury left him in a condition of Persistent Vegetative State (PSV) with 100 percent quadraplegia... Medical reports show that his medical condition has not improved in the past 13 years," LiveLaw quoted the bench as saying.
The Court noted that the continuation of his treatment -- Clinically Administered Nutrition (CAN) via surgically installed PEG tubes -- can just prolong his biological existence but will not lead to any therapeutic improvement.
What Is The Case Of Harish Rana?
Harish was a BTech student in Chandigarh who suffered severe traumatic brain injury after falling from the fourth floor of his paying guest accommodation in August 2013.
Since then, he has remained bedridden and dependent on others for all activities of daily life.
Harish's father, the petitioner, first approached the Delhi High Court in 2024, seeking permission for passive euthanasia, but was rejected as the patient was not terminally ill.
The same year, the petitioner knocked on the doors of the Supreme Court, which, though it refused to entertain the plea, directed the Uttar Pradesh government to bear the treatment expenses.
In 2025, the petitioner filed a miscellaneous application in the Supreme Court, noting that Harish's condition had no scope for improvement.
The Court then directed the constitution of a Primary Medical Board led by the District Hospital in Noida to examine his health, as well as a Secondary Medical Board constituted by the All India Institute of Medical Sciences (AIIMS).
After perusing the report, Justice Pardiwala remarked that it's a "sad report" and the man can't continue to live like this. Before passing the final order, the Court met the parents, LiveLaw reported.
The Court has asked AIIMS to provide palliative care, so that the withdrawal of CAN can be given effect to.
To maintain the dignity of death, the apex Court said that the life support must be withdrawn with a tailored plan.
1st Passive Euthanasia: What's New From The 2018 Judgment
In 2018, a five-judge Constitution Bench had recognized and given sanction for passive euthanasia, and living will/advance directives.
Later in 2023, the SC modified the guidelines, noting that withdrawal of life support is permissible only after the approval of the Primary and Secondary Medical Boards.
With the Harish Rana judgment, the apex Court today clarified how passive euthanasia should be applied in cases where a patient’s life is being supported by feeding tubes.
The top Court waived off the reconsideration period of 30 days and noted that the medical treatment, including the CAN administered to the patient, can be withdrawn or withheld.
What is Passive Euthanasia
Passive Euthanasia allows a terminally ill or irreversibly comatose patient to die naturally. It involves deliberately withholding or withdrawing life-sustaining treatments (like ventilators, feeding tubes, or medication). It has been legal since 2018, but under strict guidelines.
In Active Euthanasia, patients are administered a lethal injection to cause death. It is illegal in India and considered an offence.
The Aruna Shanbaug case in 2011 opened the door for passive euthanasia in India for the first time.
The top Court rejected euthanasia in the case of Shanbaug, a nurse at Mumbai's KEM hospital who was in a vegetative state for 42 years after an assault in 1973, as the hospital staff who cared for her for decades did not support stopping treatment.
Shanbaug continued to be under care and passed away naturally in 2015
However, in her case, the court made the judgment allowing for passive euthanasia in certain rare situations under strict conditions.
© 2024 Bennett, Coleman & Company Limited

