- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
FDA Recalls Walgreens Nasal Spray Bottles Over Contamination Risk: What You Need To Know

Credits: Canva
Medical Products Laboratories, a Philadelphia company that manufactures medical supplies, has pulled more than 41,000 bottles of Walgreens-branded nasal spray from the market after concerns that some units may contain bacteria that could pose problems for people with fragile immune systems or existing breathing issues.
The product involved is Walgreens Saline Nasal Spray with xylitol, which the U.S. Food and Drug Administration (FDA) says may be contaminated. In total, 41,328 bottles were shipped across the country and could contain pseudomonas lactis, a bacterium that naturally occurs in places such as soil and water, as noted by the Centers for Disease Control and Prevention (CDC).
Walgreens Nasal Spray Recalled: What Bacteria Is Found?
Pseudomonas lactis can cause illness in people with suppressed immunity or respiratory troubles, according to local officials in southeastern Massachusetts who reported the recall, along with television station KARE 11. WTVT also noted that the bacteria may pose added concerns for pregnant people. A 2022 study on antibiotic resistance found the same bacterium in chicken waste.
Which Walgreens Nasal Spray Lots Are Recalled?
The recall affects more than 41,000 bottles of Walgreens saline nasal spray with xylitol because of possible contamination with pseudomonas lactis, which may be risky for people who have compromised immune systems or lung conditions.
The lots included in the recall are Lot #71409, which expires on Feb. 28, 2027, and Lot #71861, which expires on Aug. 31, 2027. Walgreens told USA Today that anyone who bought these products should stop using them at once and bring them back to any Walgreens store for a complete refund.
What FDA Recall Classification Applies Here?
Based on the details available, this situation falls under a Class II recall, which the FDA uses when a product may lead to temporary or reversible health problems, and the chance of more serious harm is considered low. Similar recalls, such as the recent action involving NuNaturals sweeteners, have received the same level of risk designation from the agency.
A Class II recall signals that there is a meaningful health concern, though it is not expected to result in severe injury or death. This matches the structure of the D-0179-2026 classification and reflects how the FDA organizes consumer product recalls. A Walgreens spokesperson told FOX Business that the company’s supplier began the voluntary recall “out of an abundance of caution” and added that customer safety remains a priority.
What To Do With The Recalled Product?
Anyone who has one of the affected bottles should stop using it right away. Walgreens has not posted specific instructions on its website, but the general guidance is to discard the product or return it to the store where it was purchased.
New Zealand Launches Surveillance After Spotting Dengue, Zika-carrying Mosquito Larvae

Credit: Canva
New Zealand has stepped up surveillance after the first detection of a dengue and Zika-carrying mosquito larvae in the country.
The larval species was confirmed as 'Aedes aegypti' — known to carry diseases including dengue fever, yellow fever, Zika, and chikungunya, across the globe.
As New Zealand does not normally have the mosquito species, the larvae detected were counted as "exotic".
The mosquito species did not cause any outbreak, but were spotted during a routine surveillance program in Auckland.
"The National Public Health Service has commenced a heightened surveillance and interception programme following mosquito larvae being collected from a routine surveillance trap at Queens Wharf, Auckland, on Monday 30 March," Health New Zealand said in a statement.
Health New Zealand reported that exotic species were occasionally found at ports and airports.
Not A Public Threat Yet
The health body noted that the larvae were not considered a public health or biosecurity threat yet because there was no indication they had become established.
But the agency aimed to continue "intensive monitoring for at least three weeks".
"The monitoring would take place within a 400m radius of the site where the larvae were identified. Health Protection Officers would place mosquito traps in the survey area," the statement said.
"These have been hidden away from plain sight so they are not disturbed, for example, in old tyres, bushes, or pools of water. We ask members of the public to avoid touching or disturbing these traps if they find them, as it may disrupt our monitoring and trapping efforts," medical officer of health Dr David Sinclair said.
Sinclair said New Zealanders were most at risk from diseases transmitted by mosquitoes when travelling overseas, including to Pacific Island countries and territories where dengue fever was known to be present.
Dengue Surging In 17 Countries
The US Centers for Disease Control and Prevention recently issued a travel alert of dengue outbreaks across 17 countries.
The CDC alert issued on March 23 identified 17 countries reporting an increased number of cases of dengue. These include: Afghanistan, Bangladesh, Bolivia, Colombia, Cook Islands, Cuba, Guyana, Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, Vietnam, and the United States territories of American Samoa, Puerto Rico, and the US Virgin Islands, where local transmission is already common.
Also read: Long-term Exposure To Air Pollution Increases Fatality Rates In Dengue: Study
What Is Dengue?
Dengue is a disease caused by a virus spread through mosquito bites. It is transmitted through infected mosquitoes, primarily the species Aedes aegypti.
The breakbone fever is caused by an infection with any of four different dengue viruses. These include:
• Dengue virus type 1 (DENV-1 or DEN-1)
• Dengue virus type 2 (DENV-2 or DEN-2)
• Dengue virus type 3 (DENV-3 or DEN-3)
• Dengue virus type 4 (DENV-4 or DEN-4)
Also read: New dengue vaccine over 80% effective, prevents severe disease for up to 5 years
Common Symptoms of dengue include:
• Sudden onset of high-grade fever.
• Intense headache
• Severe muscle, joint, or bone pain.
• Skin Rash that often appears 2–5 days after the fever starts
• Nausea and Vomiting
• Minor bleeding
• Fatigue.
COVID Cicada variant: Are Children More At Risk? How To Safeguard?

Credit: iStock
The COVID Cicada variant, currently circulating in 23 countries, is likely to affect children more than the previous variants.
Children have largely escaped severe illness from the SARS-COV-2 virus.
However, the new Cicada variant with around 75 genetic changes in its spike protein is likely to disproportionately affect children, as per an expert, who noted its presence in the UK.
“Some people have done analysis on this, suggesting it may be more prevalent among young children. Children get infections all the time, but this might be something to do with the fact that they have never been exposed to Covid vaccines," Prof Ravindra Gupta, of Cambridge University, who advised the UK government during the pandemic, was quoted as saying to The Mirror.
“So this is something we’re looking at in the lab to try and work out why. The problem with this is that it is an infection that spreads fast. Eventually, it ends up in someone vulnerable," he added.
Key Symptoms
Symptoms seem to be similar to those of other recent variants and include
- sore throat,
- cough,
- congestion,
- fatigue,
- headache
- fever.
Also read: Cicada Variant: Will The Current COVID Vaccine Provide Protection?
What Do The 75 Genetic Changes Mean?
Cicada, a descendant of the Omicron variant that emerged in 2021, was first detected in South Africa in 2024. However, it disappeared soon after, only to emerge early this year.
Prof Gupta told the publication that “the Cicada variant developed the 75 mutations to escape immunity, and it evolved by incubating a single patient for over a year”.
"This 'patient zero' would have been unable to clear the virus due to a compromised immune system," due to HIV or anti-cancer drugs.
Further, he explained that due to 75 mutations to the spike protein, the body’s antibodies will take a longer time to fight the Cicada variant.
However, there is no evidence yet that it is likely to cause more severe disease.
Will The Cicada Variant Cause Severe Illness?
The Cicada variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
Yet, the World Health Organization and health experts advise that existing COVID vaccinations can help prevent severe illness and hospitalization.
"It would appear that all the protections we have from our experience with the virus and with vaccines probably offer more limited—not zero—but more limited protection against this strain," Dr. William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center, was quoted as saying by Time.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
How To Safeguard
Dr. Sai Balasubramanian, a doctor and healthcare strategy executive, writing in Forbes, stressed the need to follow COVID practices such as masks and hand hygiene.
"Healthcare professionals recommend taking general precautions, similar to those used to prevent most viral transmission: get vaccinations when appropriate, wear masks in crowded areas or indoors where there is a high risk of transmission," he said.
He also urged “avoid individuals who have known illness or infections, wash hands frequently, and continue to stay apprised of local community guidelines and the infection spread”.
India To Strictly Inspect GLP-1 Drugs To Curb Misuse: Govt Flags Risks Amid Weight-loss Hype

Credit: iStock
GLP-1 drugs are powerful tools that can improve health, but it comes with risks and caveats. While these are medical therapies, they are being misused as cosmetic products by cosmetologists, physiotherapists, dermatologists, among others.
To curb its misuse and improve public health, the Indian government today issued guidelines on its use, while also flagging risks around it.
In an official statement issued today, the government has stated that it will roll out stricter inspections to curb the misuse of GLP-1 drugs in the country and will punish those who violate the rules.
“GLP-1 drugs are a significant medical breakthrough in treating type 2 diabetes and obesity, but they are not without risk. The drugs carry a wide range of side effects - from common symptoms like nausea and vomiting to severe complications including pancreatitis, kidney injury, and bowel obstruction. These risks make it essential that GLP-1 drugs are only taken under the supervision of registered medical practitioners/specialists,” the statement said.
In the wake of its hype over weight loss, the country's regulatory authorities have taken firm steps to prevent unsupervised use and malpractices in the supply chain.
It has “strongly advised” patients and the public to” consult a qualified medical specialist before use, and to obtain these medications with prescription only through legitimate, regulated channels”.
“Stricter inspections and surveillance will continue in the coming weeks. Businesses that break the rules will face license cancellation, fines, and legal action,” the statement said.
Also read: GLP-1 Drugs: Why India Needs Stricter Rules Now
What Are The Side-effects Of GLP-1 Drugs?
GLP-1 drugs work to keep food in the digestive system for longer – making people feel fuller for longer, thus reducing appetite and inducing weight loss. Hence, the drugs are also prescribed to people with obesity.
However, when done without clinical oversight, it can lead to severe health complications.
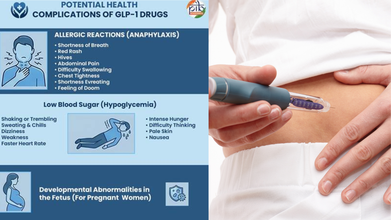
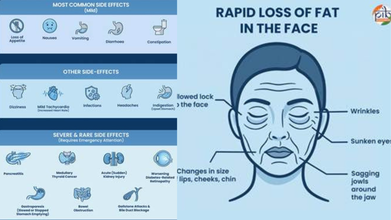
There are various side effects to taking GLP-1 drugs, including nausea and dizziness, to pancreatitis and medullary thyroid cancer.
Some serious side effects include
- Pancreatis
- Thyroid Cancer
- Acute (Sudden) Kidney Injury
- Worsening Diabetes-Related Retinopathy
- Gallstone & Bile Duct Blockage
- Rapid loss of fat in the face
- Wrinkles
- Sunken eyes
GLP-1 medications may also complicate various health conditions, such as allergic reactions, with shortness of breath and low blood sugar.
Rush For Generic Weight Loss Drugs
The government's advisory comes as patent for semaglutide -- an active ingredient in diabetes and anti-obesity drugs, specifically Wegovy and Ozempic -- expired on March 20.
This has led to a rush among Indian pharma companies to launch cheaper generic versions, significantly increasing affordability and access for millions battling Type 2 diabetes and obesity.
Also read: CDSCO threatens action against pharma companies for promoting GLP-1 weight-loss drugs
Major Indian drugmakers who launched their generic semglutide injection in the country in March include Sun Pharmaceutical Industries, Zydus Lifesciences, Alkem Laboratories, and Dr. Reddy’s Laboratories.
WHO Guidelines on GLP-1 drugs
Amid the increasing prevalence of GLP-1 drugs, the World Health Organisation (WHO), late last year, acknowledged its role in treating obesity.
However, it warned that medications like GLP-1 alone will not solve the problem affecting more than one billion people worldwide.
The global health body also issued conditional recommendations for using these therapies as part of a comprehensive approach that includes healthy diets, regular physical activity, and support from health professionals.
© 2024 Bennett, Coleman & Company Limited

