- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Heart Cells Can Regrow After A Cardiac Arrest, Scientists Say

Credit: Canva
The human heart can grow new cells in damaged areas after suffering from a heart attack, an Australian study shows.
Heart attacks occur when blood flow to the heart is severely reduced or blocked due to a buildup of plaque which is made of fat, cholesterol and other substances in the heart's arteries.
During a heart attack, a lack of blood flow causes the cells and tissue in the heart muscle to die, leading to irreversible damage that can result in serious complications like arrhythmias, heart failure, cardiogenic shock, or cardiac arrest
It is one of the leading causes of death in the country. Four Indians experience a heart attack every minute, with one in four dying of the cause. Experts have also noticed a rising trend of nearly 50 percent of heart attack patients being under the age of 40, with half of all heart attacks in Indian men occurring under 50.
However scientists in Australia have found that organ can regrow new cells to replace the damaged ones after an attack through increased mitosis.
How Does The Heart Regrow Cells?
Until now, the phenomenon of new heart cells growing in scarred areas of the heart had only been seen in mice however, the team made a breakthrough using living heart tissue samples collected from human patients undergoing bypass surgery at Australia’s Royal Prince Alfred Hospital.
The study, published in Circulation Research, found that while sections of the heart remain that had been left scarred following a heart attack, new heart muscle cells were also being produced in the same area through mitosis.
This process involves a parent cell dividing itself to create two genetically identical daughter cells, each with the same number of chromosomes as the parent for growth, repair and replacing old cells in humans.
READ MORE: The 4-Minute Scandinavian Exercise That Can Add 20 Years To Your Lifespan
Robert Hume, a research fellow at the University of Sydney and the study’s first author, said: "Until now we’ve thought that, because heart cells die after a heart attack, those areas of the heart were irreparably damaged, leaving the heart less able to pump blood to the body’s organs.
"In time, we hope to develop therapies that can amplify the heart’s natural ability to produce new cells and regenerate the heart after an attack."
The scientists are yet to discover the exact mechanism between how cells regrow in scarred regions of the heart.
Coronary Artery Disease: The Silent Killer
Coronary artery disease (CAD) is one of the most common illnesses that can cause a heart attack in people. It develops over years and has no clear signs and symptoms apart from a heart attack.
The illness begins due to a buildup of fats, cholesterol and other substances known as plaque in and on the artery walls.
Over time, this can cause narrowing or blockage of the coronary arteries and block the supply of oxygen-rich blood to heart which can lead chest pain (angina), shortness of breath and ultimately, heart attacks.
Typically, those above the age of 45, having a biological family member with heart disease, lack of sleep, smoking, consuming saturated fats along with other autoimmune diseases such as lupus and rheumatoid arthritis can increase the risk of developing CAD.
Nearly one in 10 Indian adults suffer from CAD and about two million people die from the disease annually. Apart from this, about 18 to 20 million American adults aged 20 and older are also affected about the disease.
6 Million People In Delhi Living With Poor Vision: AIIMS Study

Credit: iStock
About 6 million people in India's capital city are living with several vision problems, such as poor distance vision or near vision, according to a recent study by doctors at All India Institute of Medical Sciences (AIIMS), New Delhi.
The study, Dr. Rajendra Prasad Centre for Ophthalmic Sciences at AIIMS, and submitted to the World Health Organization (WHO), also flagged major gaps in eye care services in Delhi, the Times of India reported.
An estimated one-third of the population in the national capital was identified with refractive errors or presbyopia -- conditions that can usually be corrected with a pair of glasses. Globally, 826 million people suffer from presbyopia.
About 70 percent of older adults in the capital city were identified with the problem.
Alarmingly, about 13.1 percent school-going children had refractive errors, Dr. Praveen Vashist, professor and head of community ophthalmology at AIIMS, was quoted as saying.
Gaps In Eye Care
The study showed a lack of access to vision correction. While just 60 percent of people needing distance vision correction could access care, those with near vision correction could access care in over 47 percent of cases.
Further, the doctors found:
- women had the lowest access to spectacles compared to men
- lack of trained eye care workers,
- the affordability of spectacles,
- limited outreach to vulnerable groups -- elderly patients, rural women, and economically weaker populations.
But Dr. Vashisth noted that Delhi currently has only 1,085 ophthalmologists and approximately 489 optometrists.
Further, only 50 community-level vision centers, out of the has 249 eye care institutions in the city were found to be functioning. Moreover, only about 25 percent of children were receiving free spectacles through public health initiatives.
Global Eye Burden
According to the World Health Organization (WHO), at least 2.2 billion people worldwide have near or distance vision impairment. Of these, at least 1 billion cases could be prevented with increased access to eyeglasses and cataract surgeries
The WHO identifies refractive errors and cataracts as the leading causes of vision impairment and blindness.
Vision loss can affect people of all ages; however, most people with vision impairment and blindness are over the age of 50.
Here's how to maintain a healthy vision:
- Eat fruits and veggies, especially leafy greens like spinach, kale, and fish like salmon and tuna, to protect your sight.
- Be physically active, as being overweight or obese can increase risk for diabetes and other conditions that can lead to vision problems.
- If your vision gets blurry, use glasses to help you see better.
- Always wash your hands before putting them close to your eyes, especially if you’re putting in or taking out contact lenses.
- Use protective eyewear while playing a favorite sport or mowing the lawn
- Wear your shades to prevent the sun’s rays from hurting your eyes.
- Give your eyes a break with the 20-20-20 rule: Every 20 minutes, look about 20 feet away for 20 seconds.
- Quit smoking as it can put you at risk of serious eye issues, leading to blindness.
Metal Particle Found In Ibuprofen, Drug Now Recalled
Credits: iStock
Ibuflam tablets by pharmaceutical manufacturer Zentiva under the brand name Lichtenstein has been recalled. The 600mg tablets of ibuprofen products were recalled due to contamination. As per several media reports, a piece of metal was found in the tablet which triggered the recall. As per the manufacturer, the affected tablet are the 50-tablet packs with the batch number 5R02514.
Zentiva stated that the recall is explicitly directed at wholesalers and pharmacies, as the company confirmed upon request and the recall is not directed on patients. This measure was taken as a precautionary measure and in consultation with the relevant regulatory authorities.
As of now, patients have the option to return packages from the affected batch and receiving a replacement.
Also Read: Leucovorin Approved By FDA But For A Rare Genetic Disease, Not For Autism
How Can You Return And Receive A New Ibuprofen Prescription?
As per Zentiva, the standard procedure of returning packages from the affected batch and receiving a replacement could be availed. For instance, if the package is almost empty, the standard procedure does not need to be followed, as this could potentially be considered a new prescription. However, patients will have the option of returning packages with the batch number.
A Zentiva spokesperson explained, "The possibility of obtaining a replacement through the supplying pharmacy in case of suspected quality defects always exists. Any connection to the recall lies solely in the fact that the suspicion regarding this batch can generally be considered justified."
This is not a case of re-dispensing under the replacement procedure. It is important to note that when a replacement pack is dispensed in response to a quality complaint, the affected medicinal product is not being placed on the market again, but rather a previously prescribed and supposedly defective medicinal product is being replaced.
Read: THIS Alternative Could Help Prevent Gut Damage Caused By Painkillers
What Is Ibuflam Or Ibuprofen?
Ibuprofen is a nonsteroidal anti-inflammatory drug or NSAID that is used to relieve pain, fever, and inflammation. Ibuflam is a brand name which is a combination medication containing ibuprofen and paracetamol or acetaminophen, which is also designed for enhanced pain relief.
As per the World Health Organization (WHO), ibuprofen is on the list of essential medication. The medicine is used by mouth as a syrup or tablet, and could also be applied directly to the skin as a mousse, gel, or spray.
The uses include:
- fever
- inflammation
- headache
- menstrual pain
- common cold
- toothache
- back pain
- arthritis
- sprains
The medicine could have some side effects, which could include pain, diarrhea or constipation, nausea and vomiting, dyspepsia, bloating, dizziness, headache, nervousness, skin rash, tinnitus, edema or fluid retention.
Who Should Not Use Ibuprofen?
Anyone with:
- repeated stomach problems like heartburn
- stomach ulcers
- bleeding problems
- high blood pressure
- heart disease
- kidney disease
- age over 60 years
OR
Anyone who:
- has taken a diuretic
- is using other NSAIDs or pain relief medication
- is using anticoagulants
- is receiving treatment for any serious condition
- has chickenpox or shingles
- has Crohn's disease or ulcerative colitis
- has liver problems
Leucovorin Approved By FDA But For A Rare Genetic Disease, Not For Autism
Credits: Reuters
The US Food and Drug Administration approved leucovorin, but not for what White House had recommended for in its September briefing. FDA has approved the prescription drug leucovorin for cerebral folate deficiency in the receptor 1 gene, a genetic condition that is estimated to affect only about 1 in 1 million people.
In September, the White House touted leucovorin as a potential treatment for children with autism. Health and Me previously reported on how parents were struggling to find the prescription drug, while not enough scientific evidence was there to support the claim.
What Is Leucovorin?
It is a high-dose B vitamin, which is commonly used as a treatment to counteract the side effects of chemotherapy. It has been approved by the FDA for cerebral folate deficiency in the receptor 1 gene. The disease is rare and fewer than 50 cases have been identified worldwide. FDA Commissioner Dr Marty Makary called this approval "a significant milestone" for patients with the condition.
Also Read: Metal Particle Found In Ibuprofen, Drug Now Recalled
However, in September last year, Markary and US Health and Human Services Secretary Robert F Kennedy Jr said that the drug could help children with autism. Kennedy, standing next to US President Donald Trump, said, they had "identified an exciting therapy that may benefit large number of children who suffer from autism."
CNN reported that in Austin, Texas, Meagan Johnson, a mother with an autistic son of three years of age, spent four days calling pharmacies across the region searching for leucovorin. She contacted nearly 40 pharmacies around her home in Pflugerville, hoping to locate the medication. Health and Me reported how getting the prescription turned out to be far harder than obtaining it.
This update on leucovorin, notes CNN have hit some families hard, especially the families of children with autism. This is because of the September briefing and when Trump also said that painkiller Tylenol could in fact cause autism. The drug leucovorin was used as something that could give them hope, however, with the update in its label, parents efforts to find the prescription drugs could feel like in vain.
The Rare Condition For Which Drug Got Its Approval
The drug got its approval for folate deficiency in the receptor 1 gene. This is a defect that causes proteins that guide folate into the brain to malfunction. As a result, normal levels of folate is found in the blood, but not in the brain or nervous system, due to which tasks like thinking, speech and movement could not be performed.
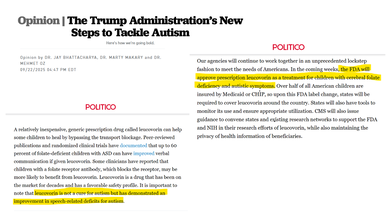
In an op/ed published in Politico and reposted on the White House website last fall, Markary said that his agency would "approve prescription leucovorin as a treatment for children with cerebral folate deficiency and autistic symptoms". However, he did not that it cause not a cure for autism, but there have been studies that show that it could help children with folate deficiency and autism to improve speech by roughly 60 per cent. The Trump administration made a bold move to expand the usage of the drug by "opening the door to the first FDA-recognized treatment for autism." wrote Markary and other HHS leaders.
© 2024 Bennett, Coleman & Company Limited

