- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
New HHS Guidelines Updates The Definitions Of Male And Female, Ignores Gender Identity

Image Credit: Canva
The U.S. Department of Health and Human Services (HHS) released new definitions for 'sex,' 'man,' and 'woman', an action that has provoked widespread discussion in health, scientific and policy communities. While proponents believe the revisions bring back biological truth, opponents caution that they will weaken science and harm healthcare policies.
On February 19, HHS Secretary Robert F. Kennedy Jr. released new federal guidelines that strictly define sex as an unchanging biological category based on reproductive function. The memo claims that sex is genetically determined at birth and does not change throughout life, making it clear that biological sex cannot be altered by medical treatment or self-identification.
The guidelines establish a female "as an individual who has a reproductive system that produces eggs (ova) and a male as an individual whose reproductive system produces sperm". The memo also indicates that hormone therapy or surgery does not change an individual's biological sex, emphasizing the view that sex is strictly determined by reproductive function. Also, it explains that sexual development disorders are not a third sex because they do not lead to the formation of a new gamete type.
These definitions will now be used in all federal agencies, affirming a binary definition of sex in federal policy and regulations. This action has been met with both approval and disapproval with those in favor believing it brings biological truth back to federal guidelines and those opposed believing it overlooks scientific complexity and may harm healthcare policies for transgender and intersex people.
Supporters of the policy, such as former HHS Office for Civil Rights Director Roger Severino, have praised the move as a return to "science and common sense." The initiative is also linked to an executive order by former President Donald Trump, which required the federal government to recognize only two sexes.
"Today, this administration is restoring common sense and biological reality to the federal government," Kennedy stated.
Its supporters contend that the former strategy, where it acknowledged gender identity in addition to biological sex, introduced uncertainty in the domains of legal protections, health coverage and data collection. They assert that affirming biological definitions will assist in upholding certainty in medical and scientific studies.
"It took a lot of years of work but we are now back to science and common sense," responded Roger Severino, the previous director of the HHS Office for Civil Rights (OCR) during the first Trump administration.
Critics Warn of Negative Health and Scientific Impacts
The move has been severely criticized by medical professionals and lobby groups, with the latter citing that the action disregards widely accepted scientific research on gender diversity. The groups argue that the policy fails to support huge bodies of research into gender identity, intersex, and how hormones affect biological sex. Through enforcing strict definitions, critics fear that the guidelines might limit transgender and nonbinary access to critical healthcare services especially those aiming at treating gender dysphoria.
Furthermore, public health research can be held back, since data collection depends on a deeper comprehension of sex and gender in order to appropriately represent diverse groups. The inability to adapt the definitions may compromise scientific progress in medical research and healthcare policies. Moreover, opponents claim that the memo overlooks the important contribution of social and psychological determinants in the development of gender identity, which have been universally acknowledged by medical societies and researchers across the globe.
How The New HHS Guidelines Might Affect Health and Healthcare
The new definitions might have significant implications across several divisions of HHS, impacting key areas like healthcare coverage, public health research, and emergency response planning.
Health Care Coverage and Affordability
Insurance companies and health centers can now be obligated to adhere to strict biological definitions of sex, with the possibility of influencing transgender healthcare service coverage, including gender-affirming treatments. This would create legal confrontations regarding medical discrimination.
Public Health and Disease Control
The epidemic studies tend to examine health hazard by gender identity as well as biological sex. If studies were limited to dual categorizations, they could frustrate attempts at comprehending as well as coping with health gaps among transgender people and nonbinaries.
Emergency Preparedness and Response
Public health interventions, such as emergency response and emergency care, typically take gender identity into account in attending to displaced or vulnerable persons' needs. Such a rigorous biological approach may truncate the utility of these efforts.
Food and Drug Safety
The FDA and regulatory agencies could have issues with approving medications that act differently according to gender identity or hormone treatment. This could affect the treatment plans for transgender individuals with diseases like osteoporosis or cardiovascular disease.
Scientific Research and Innovation
Medical and psychological studies can be impacted by limitations placed on defining gender. Funding for studies of gender identity, hormone interaction, and social determinants of health may be barred.
Helping Families and Communities
Initiatives that support non-traditional family forms, including those with transgender children or parents will be hindered if federal laws fail to acknowledge gender identity as more than an either-or differentiation.
The HHS's newly promulgated definitions of sex, man, and woman are a sweeping policy change with profound implications for healthcare, research and public health. Whereas supporters claim these definitions bring biological clarity, detractors caution they ignore scientific facts and potentially do harm to vulnerable communities. Controversy around the changes will likely persist as policymakers, medical providers and advocacy groups consider the long-term effect on health and society.
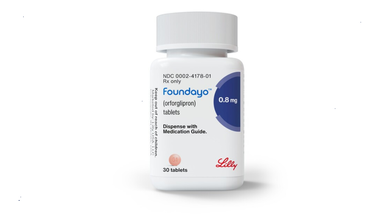
Foundayo: US FDA Approves Eli Lilly’s GLP-1 Weight Loss Pill

Credit: iStock
American drugmaker Eli Lilly’s first oral pill for weight loss, Orforglipron, marketed as Foundayo, has been given the green signal by the US Food and Drug Administration (FDA).
Lilly is also known for injectable drugs like Zepbound for obesity and Mounjaro and Trulicity for diabetes.
Foundayo’s approval comes after the US FDA in December last year approved Novo Nordisk’s Wegovy pill — the first-ever GLP-1 pill for weight loss. The Danish drugmaker rolled out the pill in January this year.
Novo Nordisk was also the first to launch oral GLP 1 Rybelsus to treat type 2 diabetes. It was approved by the US FDA in September 2019.
Also Read: World Autism Awareness Day 2026: Can Cell Therapy Be The Future Of Autism Treatment?
In a statement, the US FDA said that "Foundayo has been approved for use in combination with a reduced-calorie diet and increased physical activity. The pill is targeted to reduce excess body weight and maintain weight reduction for the long term in adults with obesity or overweight".
Lilly said that the drug will be available from April 6 through its its direct-to-consumer platform LillyDirect at a cost of $149 per month for the lowest dose for self-pay customers — on par with Novo's pill. "Shortly after” it will be available through retail pharmacies and telehealth providers in the US.
"Today, fewer than 1 in 10 people who could benefit from a GLP-1 are taking one, held back by access, stigma, perceived complexity, or the belief that their condition isn't serious enough for treatment. We believe Foundayo can help level the playing field for those living with obesity or who are overweight and living with weight-related complications," said David A. Ricks, chair and CEO of Eli Lilly and Company.
"As a convenient, once-daily oral pill that delivers meaningful weight loss, this is obesity care designed for the real world," he added.
Foundayo: A Meaningful Reduction In Body Weight
The US FDA approved Foundayo after two randomized, double-blind, placebo-controlled trials in adults with obesity or with overweight showed benefit.Also Read: Foundayo: US FDA Approves Eli Lilly’s GLP-1 Weight Loss Pill
In these trials, 72 weeks of treatment with Foundayo, in combination with a reduced-calorie diet and increased physical activity, resulted in a statistically significant and clinically meaningful reduction in body weight.
Deborah Horn, Director of the Center for Obesity Medicine at McGovern Medical School at UTHealth Houston, stated that Foundayo “delivered an average of 12.4 percent weight loss at the highest dose in clinical trials – addressing both the clinical realities of obesity and the practical challenges patients face every day."
In addition, Foundayo also led to reductions in many markers of cardiovascular risk, including waist circumference, non-HDL cholesterol, triglycerides, and systolic blood pressure across all doses, Eli Lilly said.
How Foundayo Works
Lilly licensed Orforglipron, the main ingredient in Foundayo, from a Japanese pharmaceutical company in 2018.
As with the injectable forms of GLP-1s, Foundayo is available in six doses, ranging from 0.8mg to 17.2mg.
In consultation with their doctors, patients start with the lowest dose and gradually work up to higher doses; not everyone may need to reach the highest dose.
Also read: Eli Lilly's Experimental GLP-1 Pill Shows Promising Weight Loss
Unlike the Wegovy pill, people taking orforglipron do not need to restrict food or drink after taking the pill. Orforglipron is a small molecule that the body can absorb quickly and get into the blood, where it reaches the necessary tissues.
Foundayo: Side Effects
Foundayo is not safe for use in children and has an increased list of side effects, such as tumors in the thyroid, including thyroid cancer.
The drugmaker urged watching for possible symptoms, such as
- a lump or swelling in the neck,
- hoarseness,
- trouble swallowing
- shortness of breath.
- nausea,
- constipation,
- diarrhea,
- vomiting,
- indigestion,
- stomach (abdominal) pain,
- headache,
- swollen belly,
- feeling tired,
- belching,
- heartburn,
- gas,
- hair loss.
New Zealand Launches Surveillance After Spotting Dengue, Zika-carrying Mosquito Larvae

Credit: Canva
New Zealand has stepped up surveillance after the first detection of a dengue and Zika-carrying mosquito larvae in the country.
The larval species was confirmed as 'Aedes aegypti' — known to carry diseases including dengue fever, yellow fever, Zika, and chikungunya, across the globe.
As New Zealand does not normally have the mosquito species, the larvae detected were counted as "exotic".
The mosquito species did not cause any outbreak, but were spotted during a routine surveillance program in Auckland.
"The National Public Health Service has commenced a heightened surveillance and interception programme following mosquito larvae being collected from a routine surveillance trap at Queens Wharf, Auckland, on Monday 30 March," Health New Zealand said in a statement.
Health New Zealand reported that exotic species were occasionally found at ports and airports.
Not A Public Threat Yet
The health body noted that the larvae were not considered a public health or biosecurity threat yet because there was no indication they had become established.
But the agency aimed to continue "intensive monitoring for at least three weeks".
"The monitoring would take place within a 400m radius of the site where the larvae were identified. Health Protection Officers would place mosquito traps in the survey area," the statement said.
"These have been hidden away from plain sight so they are not disturbed, for example, in old tyres, bushes, or pools of water. We ask members of the public to avoid touching or disturbing these traps if they find them, as it may disrupt our monitoring and trapping efforts," medical officer of health Dr David Sinclair said.
Sinclair said New Zealanders were most at risk from diseases transmitted by mosquitoes when travelling overseas, including to Pacific Island countries and territories where dengue fever was known to be present.
Dengue Surging In 17 Countries
The US Centers for Disease Control and Prevention recently issued a travel alert of dengue outbreaks across 17 countries.
The CDC alert issued on March 23 identified 17 countries reporting an increased number of cases of dengue. These include: Afghanistan, Bangladesh, Bolivia, Colombia, Cook Islands, Cuba, Guyana, Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, Vietnam, and the United States territories of American Samoa, Puerto Rico, and the US Virgin Islands, where local transmission is already common.
Also read: Long-term Exposure To Air Pollution Increases Fatality Rates In Dengue: Study
What Is Dengue?
Dengue is a disease caused by a virus spread through mosquito bites. It is transmitted through infected mosquitoes, primarily the species Aedes aegypti.
The breakbone fever is caused by an infection with any of four different dengue viruses. These include:
• Dengue virus type 1 (DENV-1 or DEN-1)
• Dengue virus type 2 (DENV-2 or DEN-2)
• Dengue virus type 3 (DENV-3 or DEN-3)
• Dengue virus type 4 (DENV-4 or DEN-4)
Also read: New dengue vaccine over 80% effective, prevents severe disease for up to 5 years
Common Symptoms of dengue include:
• Sudden onset of high-grade fever.
• Intense headache
• Severe muscle, joint, or bone pain.
• Skin Rash that often appears 2–5 days after the fever starts
• Nausea and Vomiting
• Minor bleeding
• Fatigue.
COVID Cicada variant: Are Children More At Risk? How To Safeguard?

Credit: iStock
The COVID Cicada variant, currently circulating in 23 countries, is likely to affect children more than the previous variants.
Children have largely escaped severe illness from the SARS-COV-2 virus.
However, the new Cicada variant with around 75 genetic changes in its spike protein is likely to disproportionately affect children, as per an expert, who noted its presence in the UK.
“Some people have done analysis on this, suggesting it may be more prevalent among young children. Children get infections all the time, but this might be something to do with the fact that they have never been exposed to Covid vaccines," Prof Ravindra Gupta, of Cambridge University, who advised the UK government during the pandemic, was quoted as saying to The Mirror.
“So this is something we’re looking at in the lab to try and work out why. The problem with this is that it is an infection that spreads fast. Eventually, it ends up in someone vulnerable," he added.
Key Symptoms
Symptoms seem to be similar to those of other recent variants and include
- sore throat,
- cough,
- congestion,
- fatigue,
- headache
- fever.
Also read: Cicada Variant: Will The Current COVID Vaccine Provide Protection?
What Do The 75 Genetic Changes Mean?
Cicada, a descendant of the Omicron variant that emerged in 2021, was first detected in South Africa in 2024. However, it disappeared soon after, only to emerge early this year.
Prof Gupta told the publication that “the Cicada variant developed the 75 mutations to escape immunity, and it evolved by incubating a single patient for over a year”.
"This 'patient zero' would have been unable to clear the virus due to a compromised immune system," due to HIV or anti-cancer drugs.
Further, he explained that due to 75 mutations to the spike protein, the body’s antibodies will take a longer time to fight the Cicada variant.
However, there is no evidence yet that it is likely to cause more severe disease.
Will The Cicada Variant Cause Severe Illness?
The Cicada variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
Yet, the World Health Organization and health experts advise that existing COVID vaccinations can help prevent severe illness and hospitalization.
"It would appear that all the protections we have from our experience with the virus and with vaccines probably offer more limited—not zero—but more limited protection against this strain," Dr. William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center, was quoted as saying by Time.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
How To Safeguard
Dr. Sai Balasubramanian, a doctor and healthcare strategy executive, writing in Forbes, stressed the need to follow COVID practices such as masks and hand hygiene.
"Healthcare professionals recommend taking general precautions, similar to those used to prevent most viral transmission: get vaccinations when appropriate, wear masks in crowded areas or indoors where there is a high risk of transmission," he said.
He also urged “avoid individuals who have known illness or infections, wash hands frequently, and continue to stay apprised of local community guidelines and the infection spread”.
© 2024 Bennett, Coleman & Company Limited

