- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Salad Dressing Recall: FDA Issues Class II Recall For Salad Dressings And Sauces Over Plastic Contamination

Credits: Canva
Salad Dressing Recall: If you recently bought salad dressing or sauces, it may be time to double-check what’s sitting in your fridge following an important recall. On November 6, the U.S. Food and Drug Administration announced a recall covering thousands of gallons of dressings and condiments made by Ventura Foods LLC. The notice was later upgraded on December 4 to a Class II recall, highlighting the need to remove these products from stores and homes. Here is what consumers should know.
Also Read: The “Triangle of Death” on Your Face: Why You Should Never Pop a Pimple There
Salad Dressing Recall: What Products Are Being Recalled?
The expanded recall affects multiple Ventura Foods LLC products, including the following dressings and sauces:
- Caesar Dressing (Costco Food Court)—32 LB (14.51 kg); SKU: 0 26700 19376 7
- Caesar Dressing (Costco Service Deli)—23.62 LB (10.71 kg); SKU: 0 26700 19376 7
- Creamy Poblano Avocado Ranch Dressing and Dip—1 GAL (3.78L); SKU: 7 34730 53243 1
- Hidden Valley, Buttermilk Ranch—1 GAL (3.79L); SKU: 0 26700 19192 3
- Italian Salad Dressing—1 GAL (3.78L); SKU: 7 67367 00518 4
- Pepper Mill Creamy Caesar Dressing—1 GAL (128 FL OZ) 3.79 L; SKU: 0 93901 78134 5
- Pepper Mill Regal Caesar Dressing—1 GAL (128 FL OZ) 3.79 L; SKU: 0 93901 72607 0
- Publix Deli Carolina-Style Mustard BBQ Sauce—12 LB; SKU: 10 026700 16964 6
- Ventura Caesar Dressing—2000LB (907.1kg); SKU: 00 026700 17360 8
Costco also recalled two ready-to-eat items that used the affected dressing: the Caesar Salad (item number 19927) and the Chicken Sandwich with Caesar Salad (item number 11444). Both products are now past their sell-by dates, which ranged from October 17 to November 9, as per Health.
Salad Dressing Recall: Why Are Salad Dressings Being Recalled?
According to the FDA and Costco, the recalled dressings and sauces may contain pieces of plastic, described as “plastic foreign material,” leading to a voluntary recall. The FDA noted that because many of these products were manufactured in large quantities for food service use, they were likely supplied to delis, cafeterias, and similar facilities. Distribution covered 27 states, including Arkansas, Colorado, Florida, Georgia, Illinois, Indiana, Kansas, Kentucky, Louisiana, Maryland, Michigan, Minnesota, Missouri, Nebraska, New Hampshire, New Jersey, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, South Carolina, Texas, Virginia, Washington, and Wisconsin.
Salad Dressing Recall: What Does A Class II Recall Mean?
On its website, the FDA explains that a Class II recall is its second most serious category. It refers to situations where using or being exposed to the product could cause temporary or medically reversible health effects, while the chance of severe health consequences is considered low.
Salad Dressing Recall: What Should I Do If I Purchased These Products?
The FDA has not issued detailed guidance for consumers who may have bought the recalled items. As with any recall, the safest step is to avoid consuming the products and contact the retailer for information about returns or refunds. Costco stated in its advisory that customers should stop using the affected items and bring them back to their local Costco location for a full refund.
3 US Hospitals Rank in the Top 10 List of World's Best Hospitals

Credits: Canva
A list by Newsweek features World's Best Hospital list in 2026. The list analyzed hospitals across 32 countries, including Australia, Austria, Belgium, Brazil, Canada, Chile, Colombia, Denmark, Finland, France, Germany, India, Israel, Italy, Japan, Malaysia, Mexico, the Netherlands, Norway, the Philippines, Saudi Arabia, Singapore, South Korea, Spain, Sweden, Switzerland, Taiwan, Thailand, Turkey, the United Arab Emirates, the United Kingdom and the United States of America.
These countries were also selected using multiple comparability criteria, which also included:
- Population size
- Life expectancy
- Standard of living
- Hospital density
- Availability of reliable data
Out of the 250 hospitals listed by Newsweek, using data from Statista, the hospitals in the United States hold four ranks in the top 10 list.
How Have The Hospitals Been Reviewed?
Each hospital was assessed and scored using four key data sources: recommendations from medical professionals—including doctors, hospital administrators and other healthcare experts—hospital quality metrics, existing patient experience data, and Statista’s Patient-Reported Outcome Measures (PROMs) Implementation Survey. PROMs are standardized, validated questionnaires completed by patients to evaluate their own functional well-being and overall quality of life.
The 2026 methodology builds on previous editions of the ranking, with Philippines and Turkey included for the first time. This year’s evaluation places greater emphasis on hospital quality metrics, integrates newly available accreditation, safety, quality and patient experience data from multiple countries, and further strengthens the importance of PROMs through increased weighting and an updated Implementation Survey.
More than 2,500 hospitals were evaluated globally, with special distinction awarded to the top 250 institutions. The five highest-ranked hospitals are: Mayo Clinic-Rochester (Rochester, Minnesota); Toronto General-University Health Network (Toronto); Cleveland Clinic (Cleveland); Karolinska Universitetssjukhuset (Stockholm); and Massachusetts General Hospital (Boston).
Access to reliable and transparent data can play a crucial role in guiding healthcare decisions. This ranking aims to serve as a practical reference for patients, families and healthcare leaders seeking trusted insight into some of the world’s most respected medical institutions.
| Rank | Hospitals | Countries |
| 1 | Mayo Clinic - Rochester | United States |
| 2 | University Health Network - Toronto General Hospital | Canada |
| 3 | Cleveland Clinic | United States |
| 4 | Karolinska Universitetssjukhuset | Sweden |
| 5 | Massachusetts General Hospital | United States |
| 6 | The Johns Hopkins Hospital | United States |
| 7 | Sheba Medical Center | Israel |
| 8 | Charité - Universitätsmedizin Berlin | Germany |
| 9 | Universitätsspital Zürich | Switzerland |
| 10 | Singapore General Hospital (SGH) | Singapore |
Where Do Indian Hospitals Rank In The List?
Of the 250 list of hospitals, India made it to the list with four hospitals. Medanta - The Medicity ranked at 110, All India Institute of Medical Sciences - Delhi ranked at 115, PGIMER - Postgraduate Institute of Medical Education and Research ranked at 214 and CMC Vellore - Town Campus at 245.
FSSAI Seized Fake 1,400 Kg of Khoya, 400 Kg of Ghee And Paneer Ahead Of Holi

Credits: FSSAI
Just ahead of Holi, the Food Safety and Standards Authority of India (FSSAI) seized fake paneer near Noida, along with 1,400 kg of fake khoya in Jhansi, and 400kg of expired ghee. The FSSAI started a series of raids in the Indian state of Uttar Pradesh and Rajasthan ahead of the festivities, when the demands of these food items are increased. Health and Me previously reported on the FSSAI raids in Kanpur where oil, rotten dates, and sweets were found to be adulterated.
Paneer Seized Near Noida At Yamuna Express Way
FSSAI on 27 February posted a video where officials could be seen disposing of a large quantity of adulterated paneer at a landfill site. FSSAI noted that the consignment was intercepted late at night at Jewar Toll Plaza on the Yamuna Expressway. The sample was seized and sent for laboratory testing, while rest of the stock was destroyed immediately.
From time and again news of adulterated paneer have made headline. Fake paneer is an adulterated, non-dairy, or low-quality substitute made using starch, hydrogenated oil (vanaspati), urea, detergent, or ammonium sulphate to mimic real milk cheese. It is found in street foods and local markets, and feels rubbery.
How to test fake paneer? The easy way to test is to use iodine on it, and if it turns blue or black, it means it is fake.
1,400 kg Of Adulterated Khoya In Jhansi
In another series of raid by FSSAI in Jhansi, officials confiscated nearly 1,400kg of adulterated khoya. This too happened as a part of a late night operation.
A mobile laboratory unit detected starch adulteration and fungal contamination in the product. The khoya confiscated was worth 3.5 lakh and was destroyed on the spot after inspectors determined that they were unsafe for consumption.
In the images shared by FSSAI, it could be clearly seen that mould-infested khoya had been dumped and discarded.
Expired Food Seized In Rajasthan
FSSAI on March 1 raided in Salumbar district of the Indian state of Rajasthan, where authorities confiscated nearly 400 kg of expired and contaminated food items. This included ghee, cooking oil, tea leaves and spices. Samples from several shops were collected and sent for tests.
Cases Of Contaminated Food In India
Not too long ago, Health and Me reported food contamination from Indore's Bhagirathpura, the same place which was the epicentre of water contamination that took many lives. Health and me also reported the news of Horlicks adulteration in Odisha.
In another news, bacteria were found in Amul milk packages, along with Mother Dairy and Country Delight. Eggoz too landed in a controversy, when a YouTube video went viral claiming that the sample tested positive for Nitrofuran.
MicrobioTx, a Bengaluru-based gut health startup tested samples from urban populations across 9 Indian states and 14 cities and found that people are significantly exposed to pesticides, insecticides, antibiotics, steroidal growth regulators, and forever chemicals.
The independent study found:
- 78% were exposed to pesticide residues, with 36% exposed to three or more pesticides, indicating cumulative exposure that may pose serious health risks.
- 54% showed presence of antibiotics, linked to antimicrobial resistance, harder-to-treat infections, and disruption of the gut microbiome that may contribute to metabolic disorders.
- 39% were exposed to steroids, compounds that may cause endocrine disruption and increased carcinogenic risk.
- 38% tested positive for forever chemicals, persistent toxins that may cause cancer risk, reduced fertility, thyroid disease, hormone suppression, high cholesterol, liver damage, and ulcerative colitis.
- 17% of the samples had 10 or more toxins in their blood across 3 categories, indicating hidden chronic exposure.
WHO Recommends Tongue Swabs For Faster Tuberculosis Diagnosis, Curbing Transmission
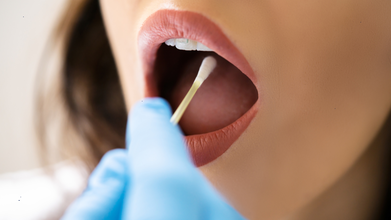
Credit: Canva
In a significant breakthrough for faster diagnosis of tuberculosis -- the world’s most infectious disease --, the World Health Organization (WHO) has recommended tongue swab tests.
The new recommendations come as many people with TB do not produce sputum, but are contributing to transmission. The swab tests may not only expand access to testing but also enable early and timely treatment that can help break chains of TB transmission.
The updated guidance also includes recommendations for the use of near point-of-care molecular tests - the nucleic acid amplification tests (NPOC-NAATs) for drug-resistant TB and pooling of sputa as a diagnostic strategy for the initial detection of TB and drug resistance.
“The WHO has just issued recommendations on new near-point-of-care (NPOC) tests for the diagnosis of #TB; easy-to-collect tongue swab samples to expand access to testing; & a cost-saving sputum pooling strategy to increase testing efficiency for TB & RR-TB,” Tereza Kasaeva, Director - WHO department on HIV, Tuberculosis, Hepatitis, in a post on social media platform X.
“These new recommendations mark a major step toward making #TB testing faster and more accessible,” she said, while calling on countries to roll out the guidelines to close diagnostic gaps.
What Are Tongue Swabs
Tongue swabs are new, readily available, and easy-to-collect specimens for use with NPOC-NAATs and low-complexity automated NAATs (LC-aNAATs) for the initial detection of TB, with and without drug resistance.
The global health body recommends using the low-complexity automated NAATs as initial diagnostic tests in adults and adolescents with signs and symptoms of lung TB.
However, in cases where respiratory samples such as sputum (expectorated or induced), tracheal aspirate, and bronchoalveolar lavage (BAL) cannot be obtained, tongue swabs may be used as initial diagnostic tests for TB.
Tongue swabs may be collected by trained personnel or self-collected with guidance from trained personnel.
Dr Rakesh PS, from the International Union Against Tuberculosis and Lung Disease (The Union), in a LinkedIn post mentioned individuals having no or minimal symptoms, and often cannot present respiratory specimens.
"Tongue swabs offer a practical way to include this otherwise “missed” group in the diagnostic pathway. By enabling testing of individuals who would otherwise be excluded, tongue swab–based testing can enhance the efficiency of active systematic screening," Dr. Rakesh said.
"Tongue swabs are a strategic alternative when obtaining a respiratory specimen is difficult or not possible -- and, when used appropriately, they can strengthen our fight against TB," he added.
What Are NPOC-NAATs
NPOC-NAATs are swab-based molecular tests for TB detection that can produce results from a primary sputum or tongue swab sample in less than one hour. These tests use instruments that can be battery-operated and do not require specialized infrastructure for use or storage.
The tests can be done in basic peripheral laboratories, such as those that perform smear microscopy, and health clinics, mobile units, or community sites that do not have laboratories.
They can be performed by health care workers with basic technical skills because they do not require laboratory methods like precision pipetting.
What Is Sputa Pooling
In sputum pooling, samples from several individuals are mixed and tested together. It is a proven strategy to improve testing efficiency and reduce costs in resource-constrained environments.
Global TB burden
While being preventable and curable, TB remains a top infectious killer, with an estimated 10.7 to 10.8 million new cases and 1.23–1.25 million deaths in 2024, as per WHO.
The infectious disease disproportionately affects low- and middle-income countries.
While TB incidence rates have shown a slight, uneven decline since 2015, the overall burden remains high, with 30 countries accounting for 87 percent of global cases, led by India, Indonesia, China, the Philippines, and Pakistan.
© 2024 Bennett, Coleman & Company Limited

