Worried About Weight Gain After Wegovy, Mounjaro? This US FDA-Approved Daily Pill May Help
Credit: Canva/Eli Lilly
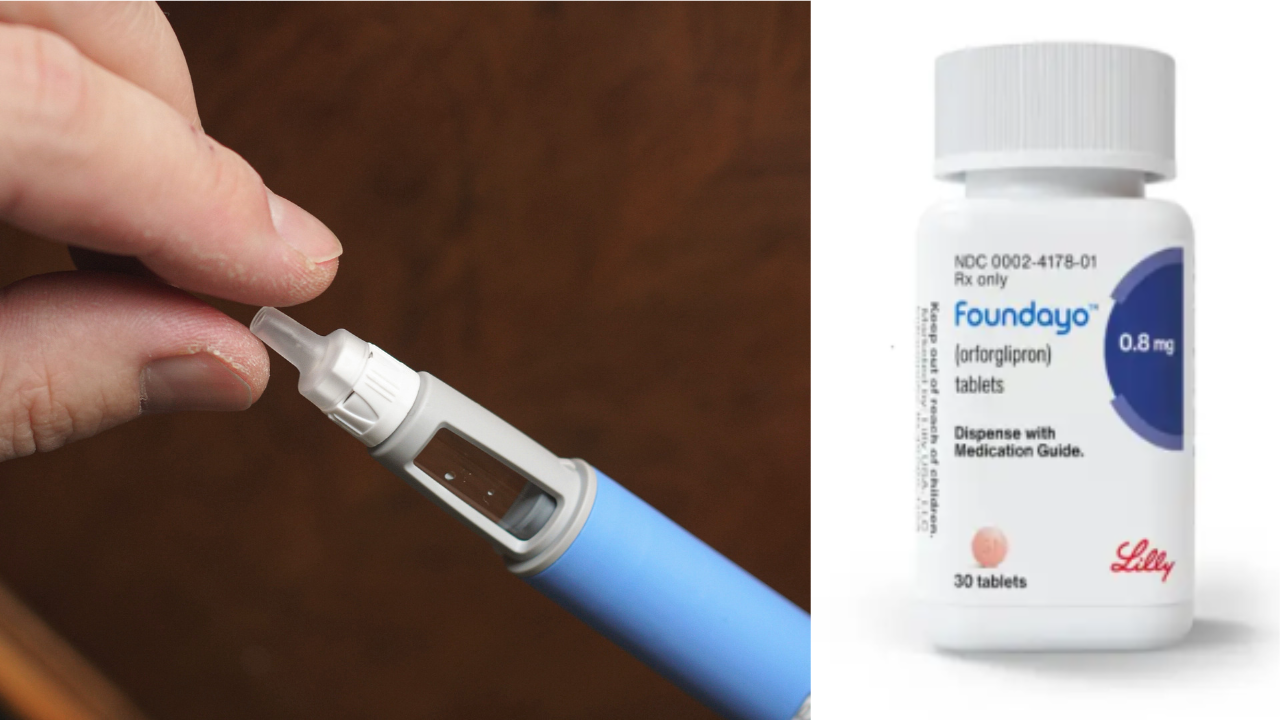
SummaryOrforglipron is US drugmaker Eli Lilly’s first oral pill for weight loss, marketed as Foundayo. In April, it received approval from the US Food and Drug Administration (FDA).
End of Article
