- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Why Has Kennedy Ordered A Review Of Baby Formula? What Every Parent Must Know About Safety

Credits: iStock
The U.S. Food and Drug Administration (FDA) is conducting a comprehensive review of infant formula ingredients. It was initiated under the guidance of Health Secretary Robert F. Kennedy Jr. as part of his "Make America Healthy Again" agenda, pledging to overhaul the U.S. food supply.
Behind this action is a mounting concern: although infant formula is still a staple for scores of American families, the science and nutritional criteria used to produce it have not materially changed since 1998. With changing worldwide research on infant nutrition and increasingly anxious parents worrying about contaminants, additives, and ingredient disclosure, this review could not be more timely.
Roughly 75% of U.S. infants are dependent on formula for the first six months, and for nearly 40%, it is the exclusive source of nutrition, as the CDC reports. In spite of world campaigns promoting exclusive breastfeeding, infant formula has emerged as a lifeline for parents who are unable—or unwilling—to breastfeed because of medical concerns, work schedules, or personal preference.
Formula is made to imitate breast milk of human women and usually comes from cow milk or soy. According to present FDA guidelines, every infant formula product is required to have 30 nutrients essential for infants in specific proportions. Yet, the technology of infants' digestion and nutrition requirements has progressed immensely, leading experts to wonder if the standards are yet sufficient.
Why Is The Government's Pushing for Nutritional Transparency?
Secretary Kennedy's call to action comes as part of a comprehensive national effort to enhance the U.S. food supply chain. The review is happening in the face of increasing parental distrust and industry trends which indicate that existing rules might not be completely in accordance with recent worldwide research on infant feeding. This effort will take into consideration an array of factors, including:
- Nutrient content and bioavailability
- Incidence of contaminants such as heavy metals
- Variations in international nutritional standards
- Possible threats from added sugars and seed oils
There is a planned roundtable discussion where scientists, manufacturers, and policymakers will engage in a debate on infant formula in the U.S. The FDA currently invites public and expert comment until the September 11 deadline.
What Are The Ingredients in Infant Formula?
Although the current formula is deemed safe, scientists insist that modern science highlights new nutritional findings not necessarily captured by existing rules. The FDA's 30-list of essential nutrients has not changed much since the 90s—even though studies on breast milk composition have dramatically changed.
Some of the main issues under consideration are:
Iron Content: American formulas generally have more iron content than European ones. Iron is crucial but, like too much of anything, could have unexpected side effects, so a rethinking may be in order.
DHA and HMOs: Docosahexaenoic acid (DHA) and human milk oligosaccharides (HMOs) are recent additions designed to make formula as much like human milk as possible. Yet these are not necessary in every product.
Added Sugars: Corn syrup solids and glucose are common in certain formulas and can be responsible for early weight gain. Lactose, the natural sugar found in breast milk, is a better option.
Seed Oils: Widely maligned by health activists, seed oils are nevertheless essential to matching the fatty acid profile of breast milk, according to experts.
Bridget Young, an infant nutritionist at the University of Rochester, pressed the point to examine beyond ingredient labels to consider bioavailability—how well the body can absorb and utilize these foods.
Kennedy's bill also seems to be in response to lessons gleaned from the 2022 formula shortage, when contamination of an Abbott plant caused a national shortage. Parents frantically sought to get safe food into their babies, demonstrating how weak and undiversified the U.S. formula supply chain really is.
More compatibility with global standards, analysts contend, would have alleviated the shortage by facilitating faster importation of safe foreign-made brands. Kennedy's proposal seeks to avoid such disruptions by increasing regulatory leeway and strengthening safety protocols.
Should Parents Be Worried?
Some parents have in the last few years opted for European infant formulas, deeming them to be more healthy because of variations in ingredient listings and the degree of labeling transparency.
Though European and American recipes vary in iron levels or forms of sugar, neither is inherently superior. Each is constructed within a distinct regulatory and cultural context.
Rather than advocating for one over the other, Kennedy's report aims to take the best from both worlds merging U.S. regulation with evidence-driven international nutritional research.
What Parents Need to Know Before Buying Baby Formula
While no short-term changes are being implemented to the amount or type of infant formula available on retail shelves, the review is a significant departure from the manner in which the federal government plans to regulate the nutrition of babies.
The review will take a minimum of one year and will require coordination among government, industry, pediatric nutritionists, and consumers. Abrams cautions that this cannot be a cosmetic effort: "No shortcuts are possible. No one white paper or committee report will suffice."
For the moment, health professionals are assuring parents that infant formulas available today are still safe and nutritionally adequate. But the future could bring formulas that are not only safe but even more biologically relevant and designed to the most recent science regarding infant development.
Maternal Vaccination During Pregnancy Can Prevent COVID-related Hospitalization In Babies: Study

Credit: iStock/Canva
Maternal vaccination with the COVID-19 vaccine during pregnancy can be effective against severe disease and hospitalization from the SARS-CoV-2 virus in babies, according to a large study.
The study, published in the journal Pediatrics, revealed that COVID vaccination during pregnancy can protect the children against hospitalization for COVID during the first six months of life.
Also Read: COVID-19 Cicada Variant: Will It Become The Dominant Strain In The US? Know All About The Virus
Amid continuing COVID cases, babies under six months old continue to have one of the highest rates of hospitalization — one in five — due to the COVID virus in the US, as per a 2024 study.
As currently no vaccines against COVID are available for neonates and babies, the American College of Obstetricians and Gynecologists (ACOG) recommends maternal vaccination during pregnancy.
Maternal COVID Vaccination Prevented Other Infections
The retrospective study included 146,031 infants born in Norway between March 2021 and December 2023. Of these, 37, 013 (25 percent) were exposed to COVID-19 vaccination in utero.
The findings showed that babies exposed to the vaccine before birth were no more likely to visit the hospital for overall infections (of any kind) than those whose mothers did not get vaccinated in pregnancy.
However, infants whose mothers were vaccinated were about half as likely to visit the hospital specifically for COVID in their first two months of life compared to babies not exposed to the vaccine in utero.
Also Read: Olivia Munn Opens Up About Detecting No-Symptom Breast Cancer With Lifetime Risk Assessment Test
Among 3 to 5-month-old babies, the risk of a hospital visit for COVID was 24 percent lower in those exposed to the vaccine, but the vaccine's protection against COVID wore off by the time infants were older than 6 months.
Importantly, the mothers' vaccine also prevented the risk of other infections in children.
"There is often an increased risk for a subsequent infection after a viral infection, such as an increased risk of pneumonia after influenza infection, so we wanted to study whether protection against COVID-19 could influence the risk of other infections as well," said lead author Dr. Helena Niemi Eide, from the University of Oslo in Norway, the NPR reported.
"But we found that COVID vaccination in pregnancy protected the infant against COVID and had no apparent effect on other infections," Eide added.
Maternal Vaccine Recommendation in the US
Last week, the American College of Obstetricians and Gynecologists reiterated its recommendation for COVID vaccination during pregnancy.
Despite changes in federal vaccine recommendations due to the US Health Secretary Robert F. Kennedy Jr.’s anti-vaccine stance, the ACOG urged COVID vaccination for
- people who are pregnant,
- recently pregnant,
- considering pregnancy,
- lactating.
Also read: Bipolar Disorder: How Early Detection Can Help Prevent Serious Complications
"Accumulated safety data from millions of administered doses show no increased risk of adverse maternal, fetal, or neonatal outcomes associated with COVID-19 vaccination in pregnancy,” the ACOG said.
'Husband Stitch': A Medical Necessity Or Just A Tool To Objectify Women's Bodies?
Credits: AI-generated and iStock
When 36-year-old mom Angela Sanford, from Fort Mill, SC, went for an appointment for a Pap smear five years after she had her first child in 2008, her nurse midwife, who she has never seen before asked her a question she did not expect. "Who stitched you up after your first birth?"
Speaking to Healthline, Sanford shared that she just started crying when the nurse said, "This is not right." Sanford said that this was the first time she ever heard the term 'husband stitch'. Sanford was told that her stich was "too tight" by the hospitalist who managed her after her first delivery.
Also Read: US CDC Warns Of New Immune-Evasive COVID Variant In 23 Countries
“He gave you what some people call a husband stitch,” Sanford recalled the midwife telling her.
“I couldn’t connect in my mind why it would be called that. My midwife said, ‘They think that some men find it more pleasurable,’” she recalled. “My husband has been worried about me and fearful of hurting me. He would never have asked for this.”
A 'Husband Stich' - What Is It?
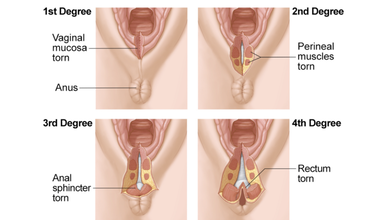
During vaginal delivery, a woman undergoes perineal tears or vaginal lacerations which means tears between the vaginal opening and anus. This causes pain, and requires stitches for grades two and higher. It also takes 4 to 6 weeks to heal. Women can experience from first to fourth degree tears.
Sometimes, a surgical incision is made in the perineum during childbirth to enlarge the vaginal opening, this is called an episiotomy. However, it is not medically necessary or a routine procedure, unless it is a case of emergency.
Stiches are required in such cases that dissolves on its own. However, a 'husband stitch', also known as "daddy stitch" is an unethical practice where an extra stitch is given during the repair process that 'tightens the vagina' to increase sexual pleasure for a male partner. While it is considered a medical malpractice, it is still done to women after vaginal delivery.
Many women face difficulty after the extra stich is given to them. In Sanford's case, she felt "excruciating" pain during sex afterwards.
Read: Romanticization And The Silent Dismissal Of Women’s Pain
When Objectifying Women's Bodies Is Made A Medical Practice
Stephanie Tillman, CNM, a certified nurse midwife at the University of Illinois at Chicago and blogger at The Feminist Midwife told Healthline: “The fact that there is even a practice called the husband stitch is a perfect example of the intersection of the objectification of women’s bodies and healthcare. As much as we try to remove the sexualization of women from appropriate obstetric care, of course the patriarchy is going to find its way in there."
Harkins, 37, said that she "kind of" laughed it off when an "old, crusty Army doctor" overstitched her so she could give her husband more pleasure. In many cases, doctors do it as a routine practice without even being told by anyone. “I couldn’t even process [it], but I kind of laughed, like what else do you do when someone says that? I had just had a baby. I didn’t think much about it because the whole birth experience was so traumatizing, but now that I think about it differently, the implications of that are just crazy.”
Episiotomy Is An Excuse For 'Husband Stitch'
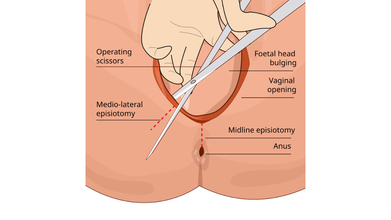
Dr Robert Barbieri, chair of obstetrics and gynecology and reproductive biology at Brigham and Women’s Hospital in Boston, told the Huffington Post that doctors were taught in the 50s and 60s that "routine episiotomy was good for women".
“What they thought is that if they did a routine episiotomy, they’d have a chance to repair it and that during the repair, they could actually create a better perineum than if they hadn’t done it. The idea [was] that we could ‘tighten things up,’” explains doctor.
However, a 2005 systematic review in the Journal of the American Medical Association found no benefit to routine episiotomy use. A 2017 Cochrane review “could not identify any benefits of routine episiotomy for the baby or the mother.” In 2016, the American College of Obstetricians and Gynecologists (ACOG) recommended that clinicians “prevent and manage” delivery lacerations through strategies like massage and warm compresses rather than making cuts on the perineum. Yet, this practice still continues inside the labor rooms.
Your Kids’ Fast-fashion Clothing May Be Laced With High Levels of Toxic Lead

Credit: iStock
While you may be buying fast-fashion clothes that are easy on your pocket and also give your children trendy looks, a new study highlights the risk of being laced with a highly toxic ingredient: lead.
The preliminary research, based on lab tests of several shirts from different retailers in the US, found that all the samples exceeded the country’s federal regulatory lead limits.
The US Consumer Product Safety Commission currently has a 100 parts per million (ppm) lead limit for children's products like toys and clothing.
"I started to see many articles about lead in clothing from fast fashion, and I realized not too many parents knew about the issue," said Kamila Deavers, principal investigator of the study, at Marian University in the US.
Deavers began the study after her young daughter’s lab reports showed elevated levels of lead in her blood from toy coatings.
How was study conducted?
The team tested 11 shirts that spanned the rainbow—red, pink, orange, yellow, gray, and blue. All brightly colored fabrics, particularly reds and yellows, showed higher levels of lead compared to more muted tones.
"We saw that the shirts we tested were all over the allowed limit for lead of 100 ppm," said Priscila Espinoza, from Marina.
The researchers explained that some manufacturers use lead (II) acetate as an inexpensive way to help dyes stick to the materials and produce bright, long-lasting color.
Children At Greater Risk
The researchers found the risk is particularly higher among younger kids as they tend to playfully suck or chew their clothes during play.
"Even briefly chewing these fabrics could expose children to dangerous lead levels,” they found in the study, to be presented at the forthcoming meeting of the American Chemical Society.
According to the US Environmental Protection Agency (EPA), lead exposure can lead to behavior problems, brain and central nervous system damage, as well as other negative health effects in children.
The agency considers children under six years old to be most at risk from exposure. Even low levels of lead in the blood of children can result in:
- Behavior and learning problems
- Lower IQ and Hyperactivity
- Slowed growth
- Hearing Problems
- Anemia
The researchers also pointed out safer alternatives to lead-based dyeing agents that already exist. These include natural and less harmful substances such as:
- Plant-based tannins (e.g., oak bark, pomegranate peel)
- Rosemary
- Alum.
© 2024 Bennett, Coleman & Company Limited

