- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
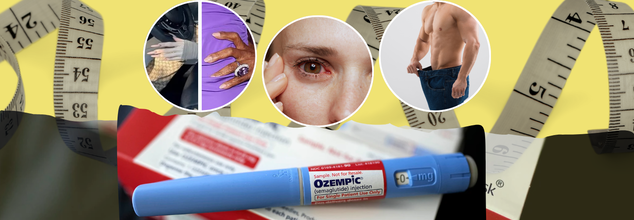
Bizarre Penis Changes, Eye Strokes, Creepy ‘Walking Dead’ Hands: Alarming Ozempic Side Effects Shocking The World
Credits: Health and me
By now most people are familiar with the popular weight loss injection Ozempic—the so-called miracle shot for weight loss that's got everyone talking, from celebs to your next-door neighbor. But while the number on the scale might be dropping, something else is happening too… and it’s a little bizarre.
Ever scrolled through TikTok and stumbled on people talking about “Ozempic hands”? Or read wild claims about sudden growth in places you didn’t expect? Yep, that’s all part of it. And just when you thought it couldn’t get weirder—there are whispers about vision loss, creepy-looking fingers, and even “walking dead” hands. Sure, Ozempic helps you shed pounds fast. But at what cost?
If you’ve been curious, concerned, or just downright confused about what this drug is really doing behind the scenes, buckle up—we’re diving into the strangest, scariest, and most surprising side effects people are reporting. Let’s talk about what no one told you before you got the jab.
Once hailed as a miracle for weight loss and diabetes management, Ozempic (semaglutide) is now under scrutiny for a growing list of bizarre and disturbing side effects. While the drug continues to dominate the market for its ability to help people shed up to 20% of their body weight in a year, disturbing new reports have surfaced. From surprising anatomical changes to eye strokes and skeletal hands, the downsides of this blockbuster injection are no longer whispered rumors—they're headlining forums, social media, and even regulatory reports.
Also Read: Ozempic Vulva To Ozempic Smell – Unexpected Ways Of Weight Loss Jabs Change Your Body
Penis Enlargement or Illusion?
One of the most viral and eyebrow-raising claims is the supposed enlargement of the penis in men taking Ozempic. Reddit threads have buzzed with firsthand accounts of users noticing visible growth. One user wrote, "I recently measured myself down there and noticed I gained about one inch."
While there is no solid scientific study directly linking semaglutide to penile growth, experts suspect the phenomenon may be tied more to fat loss than actual tissue increase. As men lose significant weight—especially in the pubic region—the buried portion of the penis becomes more visible, creating the illusion of increased size. Still, the global trend is puzzling: the UK saw average sizes increase from 5.17 to 5.63 inches between 2022 and 2024, with Venezuelan men reporting an even more staggering jump from 1.42 to 6.67 inches.
While these figures are alarming, the truth may be buried in overlapping factors like obesity reduction, changes in prostate health, and increased sexual function—rather than Ozempic alone.
Eye Strokes and Irreversible Eye Damage
What may be even more serious than Reddit threads is what regulators are now confirming: Ozempic may cause a rare yet dangerous eye condition known as non-arteritic anterior ischemic optic neuropathy (NAION). The European Medicines Agency (EMA) recently asked Novo Nordisk, Ozempic’s manufacturer, to list NAION as a rare side effect in their drug information.
The condition is the second most common cause of vision loss due to optic nerve damage, trailing only glaucoma. It is believed to affect as many as 1 in 10,000 users of semaglutide after a year of use. Alarmingly, a 2023 study involving nearly 350,000 diabetic patients found that those using semaglutide had more than double the risk of developing NAION.
While Novo Nordisk maintains that its drug's benefit-risk profile remains favorable, doctors and analysts warn that continued monitoring and patient awareness are critical.
Ozempic Hands, Feet, Face
Beyond internal risks, Ozempic is transforming bodies in eerie, visible ways. Welcome to the world of "Ozempic hands," "Ozempic feet," and "Ozempic face."
Ozempic hands refer to the skeletal, aged appearance some users develop due to rapid subcutaneous fat loss in the fingers and palms. This phenomenon gained traction after several viral TikTok videos and celebrity photos pointed out the dramatically thinned hands of public figures rumored to be on weight-loss jabs.

People report needing to resize wedding rings, and in some cases, cosmetic procedures like hand fillers are being considered to combat the bony transformation. The same process is evident in the feet, with sagging skin and even discomfort in walking being reported.
Experts explain that these injections don’t just target fat in one part of your body. When you lose weight this quickly, your face, your hands, your feet—everything shows it.
The "melting candle" look is another post-weight loss dilemma facing Ozempic users. With such dramatic fat loss, many people are left with drooping excess skin that doesn’t snap back. Surgeons report a boom in body contouring surgeries such as tummy tucks, thigh lifts, and arm lifts.
"It’s not just vanity," says one bariatric surgeon. "Excess skin can lead to hygiene issues, infections, and mobility challenges."
In many cases, the only solution is surgery—an expensive, invasive, and sometimes risky route. Procedures can cost thousands of dollars and carry their own set of complications, including blood clots and infection.
Ozempic and its sister drugs like Wegovy and Mounjaro are rapidly becoming household names. In the UK alone, over 350,000 people are now prescribed GLP-1 receptor agonists, with another half a million believed to be using them privately. In the US, about 13% of the population—around 33 million people—have tried the injections.
With newer policies allowing GPs in the UK to prescribe these drugs without requiring specialist consultations, usage is expected to soar. But so too are concerns about long-term side effects.
There’s no denying that Ozempic and related medications have transformed lives by aiding weight loss and managing type 2 diabetes. Many users experience improved cardiovascular outcomes, better blood sugar control, and renewed energy but as the side effects multiply—ranging from the bizarre (penis changes) to the debilitating (eye strokes and excess skin)—patients and healthcare providers must weigh these drugs' promises against their perils.
US Woman In Active Labor Forced to Face Court Over Refusal Of C-Section
Credit: ProPublica
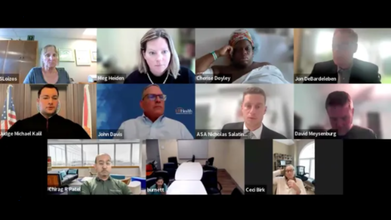
In a disturbing case from the US state of Florida, a pregnant woman in active labor was forced to attend a virtual court hearing via Zoom from her hospital bed for refusing a Cesarean delivery, also known as C-section — a common method of childbirth.
ProPublica reported that Cherise Doyley was in her 12th hour of contractions at the University of Florida Health facility, when she was, without her consent, made to sit in front of a host of people — a judge in a black robe and several lawyers, doctors, and hospital staff — for the Zoom proceeding.
While in active labor, a nurse came in with a bedsheet and told her to cover up, and a supervisor followed with a tablet.
“It’s a real judge in there?” Doyley asked the nurse at the beginning of what would be a three-hour hearing.
What Was The Case?
The mother of three, and a professional birthing doula, Doyley had arrived at the facility after her water broke.
While her doctors expressed concerns about the risk of uterine rupture —a potentially deadly complication for her and her baby — Doyley wanted to try for a vaginal delivery, as the risk was less than 2 percent, unless there was an emergency.
She told doctors she wouldn’t consent to a cesarean without trying to have a vaginal delivery first.
While the doctors initially relented, after several hours, she had to face a virtual court hearing, where the hospital and state attorney’s office forced Doyley to undergo a cesarean section.
Doyley has her own reasons to avoid a C-sec. She already had three prior C-sections, and one that resulted in a hemorrhage. She feared that a C-sec would lead to another serious complication and a lengthy recovery, and her kids would suffer.
However, the hospital was worried that her medical decisions may cause harm to the fetus, and that the courts may help decide which one mattered more, the report said.
Also read: Maternal Vaccination During Pregnancy Can Prevent COVID-related Hospitalization In Babies: Study
After three hours of testimony — all while Doyley lay in her hospital bed — the judge ruled that she could keep laboring unless there was an emergency. If that happened, the hospital could operate, whether she wanted it or not.
Overnight, doctors said the baby’s heart rate dropped for seven minutes. Doyley woke to her hospital bed being wheeled into surgery. The baby girl was delivered by C-section, the report said.
What Happens In A C-section?
It is a surgical procedure that is used to deliver a baby through an incision made in the abdomen and uterus.
The method is preferred, especially in cases with complications during labor or breech presentation, or multiple births.
Unlike the popular conception, it hurts. In a vaginal delivery, the pain is experienced during labor and pushing, especially if done without an epidural.
Whereas, in a C-section, a surgery is performed that numbs the body from the chest down. However, the recovery could be painful and prolonged. It is also because in a C-section, it involves healing from a major abdominal surgery, while vaginal birth recovery may be quicker, more complicated, or traumatic.
Also read: 'Husband Stitch': A Medical Necessity Or Just A Tool To Objectify Women's Bodies?
Does A C-sec Affect The Baby?
Usually, it does not harm the baby, but there could be potential risks to the baby, including:
- Surgical injury, which is very rare; however, the baby may be accidentally nicked during the incision.
- Respiratory issues: Babies born via C-section may have a higher risk of breathing, since they miss the natural compression during the vaginal birth that helps expel the amniotic fluid from their lungs
- Delayed bonding: Immediate skin-to-skin contact may take longer due to the surgical process.
- Feeding challenges: Some newborns may initially struggle with breastfeeding.
Risks Of A C-section Birth?
- Excessive blood loss
- Infection
- Blood clots in the legs, lungs, or pelvis
- Damage to nearby organs
- Adverse reactions to anesthesia
The American College of Obstetricians and Gynecologists (ACOG) advises against elective C-sections due to these potential complications.
Foundayo: US FDA Approves Eli Lilly’s GLP-1 Weight Loss Pill

Credit: iStock
American drugmaker Eli Lilly’s first oral pill for weight loss, Orforglipron, marketed as Foundayo, has been given the green signal by the US Food and Drug Administration (FDA).
Lilly is also known for injectable drugs like Zepbound for obesity and Mounjaro and Trulicity for diabetes.
Foundayo’s approval comes after the US FDA in December last year approved Novo Nordisk’s Wegovy pill — the first-ever GLP-1 pill for weight loss. The Danish drugmaker rolled out the pill in January this year.
Novo Nordisk was also the first to launch oral GLP 1 Rybelsus to treat type 2 diabetes. It was approved by the US FDA in September 2019.
Also Read: World Autism Awareness Day 2026: Can Cell Therapy Be The Future Of Autism Treatment?
In a statement, the US FDA said that "Foundayo has been approved for use in combination with a reduced-calorie diet and increased physical activity. The pill is targeted to reduce excess body weight and maintain weight reduction for the long term in adults with obesity or overweight".
Lilly said that the drug will be available from April 6 through its its direct-to-consumer platform LillyDirect at a cost of $149 per month for the lowest dose for self-pay customers — on par with Novo's pill. "Shortly after” it will be available through retail pharmacies and telehealth providers in the US.
"Today, fewer than 1 in 10 people who could benefit from a GLP-1 are taking one, held back by access, stigma, perceived complexity, or the belief that their condition isn't serious enough for treatment. We believe Foundayo can help level the playing field for those living with obesity or who are overweight and living with weight-related complications," said David A. Ricks, chair and CEO of Eli Lilly and Company.
"As a convenient, once-daily oral pill that delivers meaningful weight loss, this is obesity care designed for the real world," he added.
Foundayo: A Meaningful Reduction In Body Weight
The US FDA approved Foundayo after two randomized, double-blind, placebo-controlled trials in adults with obesity or with overweight showed benefit.Also Read: Foundayo: US FDA Approves Eli Lilly’s GLP-1 Weight Loss Pill
In these trials, 72 weeks of treatment with Foundayo, in combination with a reduced-calorie diet and increased physical activity, resulted in a statistically significant and clinically meaningful reduction in body weight.
Deborah Horn, Director of the Center for Obesity Medicine at McGovern Medical School at UTHealth Houston, stated that Foundayo “delivered an average of 12.4 percent weight loss at the highest dose in clinical trials – addressing both the clinical realities of obesity and the practical challenges patients face every day."
In addition, Foundayo also led to reductions in many markers of cardiovascular risk, including waist circumference, non-HDL cholesterol, triglycerides, and systolic blood pressure across all doses, Eli Lilly said.
How Foundayo Works
Lilly licensed Orforglipron, the main ingredient in Foundayo, from a Japanese pharmaceutical company in 2018.
As with the injectable forms of GLP-1s, Foundayo is available in six doses, ranging from 0.8mg to 17.2mg.
In consultation with their doctors, patients start with the lowest dose and gradually work up to higher doses; not everyone may need to reach the highest dose.
Also read: Eli Lilly's Experimental GLP-1 Pill Shows Promising Weight Loss
Unlike the Wegovy pill, people taking orforglipron do not need to restrict food or drink after taking the pill. Orforglipron is a small molecule that the body can absorb quickly and get into the blood, where it reaches the necessary tissues.
Foundayo: Side Effects
Foundayo is not safe for use in children and has an increased list of side effects, such as tumors in the thyroid, including thyroid cancer.
The drugmaker urged watching for possible symptoms, such as
- a lump or swelling in the neck,
- hoarseness,
- trouble swallowing
- shortness of breath.
- nausea,
- constipation,
- diarrhea,
- vomiting,
- indigestion,
- stomach (abdominal) pain,
- headache,
- swollen belly,
- feeling tired,
- belching,
- heartburn,
- gas,
- hair loss.
New Zealand Launches Surveillance After Spotting Dengue, Zika-carrying Mosquito Larvae

Credit: Canva
New Zealand has stepped up surveillance after the first detection of a dengue and Zika-carrying mosquito larvae in the country.
The larval species was confirmed as 'Aedes aegypti' — known to carry diseases including dengue fever, yellow fever, Zika, and chikungunya, across the globe.
As New Zealand does not normally have the mosquito species, the larvae detected were counted as "exotic".
The mosquito species did not cause any outbreak, but were spotted during a routine surveillance program in Auckland.
"The National Public Health Service has commenced a heightened surveillance and interception programme following mosquito larvae being collected from a routine surveillance trap at Queens Wharf, Auckland, on Monday 30 March," Health New Zealand said in a statement.
Health New Zealand reported that exotic species were occasionally found at ports and airports.
Not A Public Threat Yet
The health body noted that the larvae were not considered a public health or biosecurity threat yet because there was no indication they had become established.
But the agency aimed to continue "intensive monitoring for at least three weeks".
"The monitoring would take place within a 400m radius of the site where the larvae were identified. Health Protection Officers would place mosquito traps in the survey area," the statement said.
"These have been hidden away from plain sight so they are not disturbed, for example, in old tyres, bushes, or pools of water. We ask members of the public to avoid touching or disturbing these traps if they find them, as it may disrupt our monitoring and trapping efforts," medical officer of health Dr David Sinclair said.
Sinclair said New Zealanders were most at risk from diseases transmitted by mosquitoes when travelling overseas, including to Pacific Island countries and territories where dengue fever was known to be present.
Dengue Surging In 17 Countries
The US Centers for Disease Control and Prevention recently issued a travel alert of dengue outbreaks across 17 countries.
The CDC alert issued on March 23 identified 17 countries reporting an increased number of cases of dengue. These include: Afghanistan, Bangladesh, Bolivia, Colombia, Cook Islands, Cuba, Guyana, Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, Vietnam, and the United States territories of American Samoa, Puerto Rico, and the US Virgin Islands, where local transmission is already common.
Also read: Long-term Exposure To Air Pollution Increases Fatality Rates In Dengue: Study
What Is Dengue?
Dengue is a disease caused by a virus spread through mosquito bites. It is transmitted through infected mosquitoes, primarily the species Aedes aegypti.
The breakbone fever is caused by an infection with any of four different dengue viruses. These include:
• Dengue virus type 1 (DENV-1 or DEN-1)
• Dengue virus type 2 (DENV-2 or DEN-2)
• Dengue virus type 3 (DENV-3 or DEN-3)
• Dengue virus type 4 (DENV-4 or DEN-4)
Also read: New dengue vaccine over 80% effective, prevents severe disease for up to 5 years
Common Symptoms of dengue include:
• Sudden onset of high-grade fever.
• Intense headache
• Severe muscle, joint, or bone pain.
• Skin Rash that often appears 2–5 days after the fever starts
• Nausea and Vomiting
• Minor bleeding
• Fatigue.
© 2024 Bennett, Coleman & Company Limited

