- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Delhi Sees Rising Cancer Burden With Sharp Increase in Oral and Lung Cancers; Breast Cancer Remains Most Common

Credits: iStock and Canva
Oral cancer cases recorded the sharpest in Delhi in 2025, as per the Union Health Ministry data presented in the Parliament. While there is no definitive explanation provided by the report by the Union Health Ministry, previous reports show that widespread use of tobacco has increased, and accounted for about 30% of all oral cancer cases in India alone.
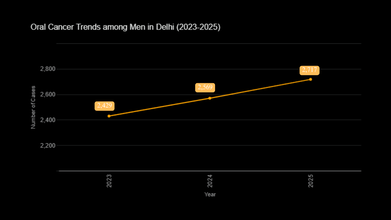
As per the report shared by the Union Health Ministry, a 5.1% increase was scene in the reported increase of oral cancer, while lung cancer rose by 4.9% between the period 2023 and 2025.
Also Read: ‘Fix Your Handwriting’: Indian Doctors Told to Write Clear, Legible Prescriptions Under New Rules
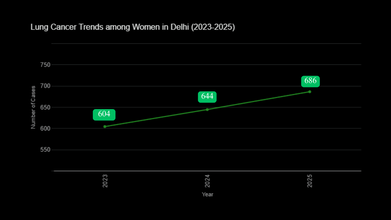
The data showed that lung cancer increased among women, and it rose to 6.5%. In numbers, this would make 686 cases in 2025, which is up from the 644 cases recorded in 2024, and 604 in 2023. Among men, oral cancer had the highest growth, with a 5.8% increase. This led to 2,717 cases in 2025, compared to 2,569 cases in 2024 and 2,429 cases in 2023.
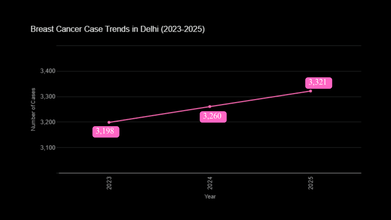
However, breast cancer remained the most prevalent cancer in the city. Data from the National Cancer Registry Programme indicate a steady rise in reported breast cancer cases, increasing from 3,198 in 2023 to 3,260 in 2024 and 3,321 in 2025. Oral cancer ranked as the second most common cancer overall, with combined cases among men and women reaching 3,208 in 2025.

A closer look at the data shows that oral cancer continued to rise in the city over the past three years. In 2023, Delhi reported 2,901 cases, including 2,429 among men and 472 among women. The number rose to 3,051 cases in 2024, with 2,569 cases in men and 482 in women. Among women, cases increased slightly further to 491 in 2025.
The International Journal of Research in Medical Sciences provide the tobacco use, betel quid chewing has seen a rising trend in India. Tobacco use includes chewing gutka and khaini in rural areas as well as smoking cigarettes in urban area, which continues to be the nation's leading preventable cause of cancer.
Cervical Cancer Shows a Decline
Cervical cancer followed a different pattern. Cases among women fell from 741 in 2023 to 716 in 2024, and then to 692 in 2025. This reflects a year-on-year decline of around 3.4 percent over both periods.
Rising Trends in Other Cancers Among Men
Among men, oral cancer showed the sharpest increase. Lung cancer cases also rose, climbing from 1,668 in 2023 to 1,814 in 2025. Prostate cancer followed a similar upward trend, with reported cases increasing from 1,168 in 2023 to 1,301 in 2025.
Delhi’s High Cancer Incidence Rate
The health ministry has pointed to Delhi’s relatively high Age-Adjusted Incidence Rate of cancer. Between 2015 and 2019, the AAIR stood at 146.7 per one lakh population among men and 132.5 among women. These figures are higher than those reported in cities such as Mumbai, Kolkata, Pune and Ahmedabad. The AAIR allows cancer rates to be compared fairly across regions with different age profiles.
Treatment Burden Extends Beyond Delhi
It is also important to note that Delhi attracts a large number of cancer patients from across north India and other parts of the country. Major tertiary care centres, including AIIMS and the Rajiv Gandhi Cancer Institute and Research Centre, contribute to a higher treatment load in the city.
Kent meningitis outbreak climbs to 27 cases, UK officials warn numbers may continue to rise

Credit: Canva
UK health officials have identified seven more cases of meningitis, as part of the latest outbreak in Kent, taking the total number of cases to 27. They expect the numbers to rise in the coming days.
The UK Health Security Agency noted that it has expanded the Meningitis B vaccine regimen to everyone who has been offered preventative antibiotic treatment as part of this outbreak.
“15 laboratory cases are confirmed and 12 notifications remain under investigation, bringing the total to 27,” the UKHSA said in a statement.
“Currently, cases have been confirmed in students at 4 schools in Kent, as well as one student at a higher education institution in London (who is confirmed to be directly linked to the outbreak),” it added.
Two students -- a 21-year-old student at the University of Kent and a teenage student at a school in the town of Faversham have died in the outbreak.
A 9-month-old baby from Folkestone is reportedly battling for life in the intensive care unit.
Meanwhile, another university in the city confirmed a case of meningitis. The student at Canterbury Christ Church is believed to be a man who was at the nightclub and part of the initial cluster of 20 known cases, who visited Club Chemistry in Canterbury between March 5 and 7.
"We are not in the position yet to say definitively that it's been contained," Dr Anjan Ghosh, Director of Public Health at Kent County Council, told BBC Radio, adding that secondary transmissions needed to be ruled out.
Calling the outbreak “unprecedented”, Health Minister Wes Streeting said: “The number of suspected cases was expected to increase in the coming days because the disease had a seven- to 10-day incubation period”, Reuters reported.
In a typical year, Britain sees about 350 cases, roughly one per day, according to government estimates.
In addition to the approximately 5,000 students who were initially contacted, vaccination will now be extended to everyone who has been offered preventative antibiotic treatment as part of this outbreak, the UKHSA said.
Why The Cases Are Rising
While it remains unclear why the outbreak has been so large, “the large number of cases all originating from what seems to be a single event” is particularly striking, Prof Robin May, the chief scientific officer at the UKHSA, told the BBC Breakfast.
May said that "there might be something about the kind of behaviors that individual people are doing." Another probable reason "is that the bacteria may have evolved to be better at transmitting".
Both the UKHSA and the European Centre for Disease Prevention and Control maintain that the risk of invasive meningococcal disease to the general population in Europe is "very low".
Also read: From Vaping to Worm Attack: 5 Unimaginable Ways to Contract Meningitis
Can The Outbreak Be Prevented?
The UKHSA noted that the key intervention to protect people and halt the spread remains for people to come forward for antibiotic treatment -- effective in preventing contraction and spreading in 90 percent of cases.
In addition, a targeted MenB vaccination program is also being introduced for longer-term protection.
“By extending the vaccination program to everyone who has been offered preventative antibiotics, we are taking an important additional step to protect those most likely to have been exposed. The message is simple: if you have had the antibiotic, you are also eligible for the vaccination,” said Professor Susan Hopkins, Chief Executive of the UKHSA.
However, Trish Mannes, UKHSA Regional Deputy Director for the South East, noted that even after two doses, the MenB vaccine “does not protect against all strains of meningococcal disease, nor against all infections that can cause meningitis. It also does not prevent the bacteria from being carried and spread in the community”.
The UKHSA thus warned people to be aware of the signs and symptoms of invasive meningococcal disease, and to seek immediate medical attention if they or anyone they know develops these signs and symptoms.
Common symptoms include:
- rash
- sudden onset of high fever
- severe and worsening headache
- vomiting and diarrhoea
- joint and muscle pain
- seizures.
Study Links Widespread Use of Antibiotics During COVID To Surge In AMR Cases

Credit: iStock
The widespread use of Azithromycin to treat hospitalized patients during the COVID-19 pandemic increased the risk of antimicrobial resistance -- a major global health problem, according to a new study, published in the journal Nature Microbiology.
Scientists at the University of California-San Francisco (UCSF) noted that using azithromycin inappropriately for even a single day can trigger antibiotic resistance in the respiratory tract.
While azithromycin is effective against bacterial infections that cause strep throat, pneumonia, and sexually transmitted diseases, it does not work against viruses.
"We've known for years that antibiotics don't treat viral infections, but these results were striking," said Chaz Langelier, from UCSF.
"That we could see resistance genes turning on in the respiratory tract within a day tells us the consequences of unnecessary antibiotic use aren't theoretical or long-term. They're immediate, measurable, and biologically real," Langelier added.
The study analyzed nasal swabs of 1,164 adults hospitalized for COVID-19 to examine the changes that occurred in the microbiome of hospitalized patients who were treated for COVID.
Compared to people who received no antibiotics, patients administered azithromycin reported changes that persisted for more than a week. These include:
- Changes in the mix of microbes in the upper airway
- Decrease in harmless bacteria,
- Surge in potentially harmful bacteria such as Staphylococcus and Klebsiella.
Rising Global Antimicrobial Resistance
Antimicrobial resistance (AMR) occurs when germs develop the ability to defeat the drugs designed to kill them.
It is one of the 10 top global health threats, undermining the effectiveness of essential treatments and placing millions at risk of untreatable infections.
As per WHO data, AMR is an urgent global public health threat, killing at least 1.27 million people worldwide and associated with nearly 5 million deaths in 2019.
In the US alone, more than 2.8 million antimicrobial-resistant infections occur each year. More than 35,000 people die as a result, according to the CDC's 2019 Antibiotic Resistance (AR) Threats Report.
The WHO, in a 2025 report, noted that one in six laboratory-confirmed bacterial infections causing common infections in people worldwide in 2023 were resistant to antibiotic treatments.
Between 2018 and 2023, antibiotic resistance rose in over 40 percent of the monitored antibiotics with an average annual increase of 5-15 percent.
US Early Death Toll During COVID Much Higher
About 16 per cent of COVID-19 deaths went uncounted early in the pandemic in the US, according to a separate study, published by the journal Science Advances.
While about 840,000 COVID deaths were reported on death certificates in 2020 and 2021, the researchers using artificial intelligence (AI) decoded that as many as 155,000 unrecognised additional deaths likely occurred in that time outside of hospitals.
Icotyde: US FDA Approves Johnson & Johnson's Pill For Psoriasis
Credit: J&J/Canva
The US Food and Drug Administration has approved Johnson & Johnson's once-daily pill for psoriasis -- a chronic skin disease.
Icotyde is an interleukin-23 (IL-23) receptor antagonist approved for the treatment of moderate-to-severe plaque psoriasis in adults and pediatric patients 12 years of age and older.
Psoriasis is an autoimmune condition that causes rough patches of skin. The new once-daily pill has been found safe and also delivers complete skin clearance.
Also Read: Why Regulatory Clarity Is Important for Safe Aesthetic Procedures in India
In the statement, J&J said that the approval will lead to a first-line systemic treatment of plaque psoriasis with a targeted oral peptide.
“Icotyde delivers something unique in psoriasis treatment – combining skin clearance with a favorable safety profile in a once‑daily pill, making it an easy addition to a patient’s routine,” said Linda Stein Gold, Director of Dermatology Clinical Research at Henry Ford Health.
The novel pill also aligns with the recent International Psoriasis Council guidance that suggests transitioning to systemic therapy if two cycles of topical medications applied for four weeks fail to bring meaningful improvement.
Icotyde will prove to be “a potential game‑changer for many adult and adolescent patients”, Gold said.
The FDA’s approval is based on an unprecedented body of evidence from the phase three clinical trial, which simultaneously evaluated Icotyde in adults and adolescents, and found it a safe and effective oral pill, and also better than injectables and topical creams.
According to analysts from Wall Street, Icotyde has "blockbuster potential," and the once-daily medication could capture significant market share from injectables like Skyrizi and J&J's own Tremfya, Reuters reported.
J&J is also studying the drug, chemically known as icotrokinra, for ulcerative colitis, psoriatic arthritis, and Crohn's disease.
What Is Psoriasis?
Psoriasis develops when the body makes skin cells too quickly. It causes skin cells to pile up and form visible patches or spots on the skin, which may be itchy or painful.
The condition, however, is not contagious.
According to the National Psoriasis Foundation, the skin disease affects 8 million Americans and more than 125 million people worldwide.
The condition impacts physical comfort and quality of life, especially when lesions are on visible or sensitive areas.
The plaques typically appear as raised patches with a silvery white buildup of dead skin cells or scales. They can appear anywhere on the body, although they most often appear on the scalp, knees, elbows, and torso.
Who Can Use Icotyde
Icotyde is currently approved in the US for the treatment of people with moderate-to-severe plaque. It can be used by
- adults, and pediatric patients 12 years of age and older
- who weigh at least 40 kg.
© 2024 Bennett, Coleman & Company Limited

