Icotyde: US FDA Approves Johnson & Johnson's Pill For Psoriasis
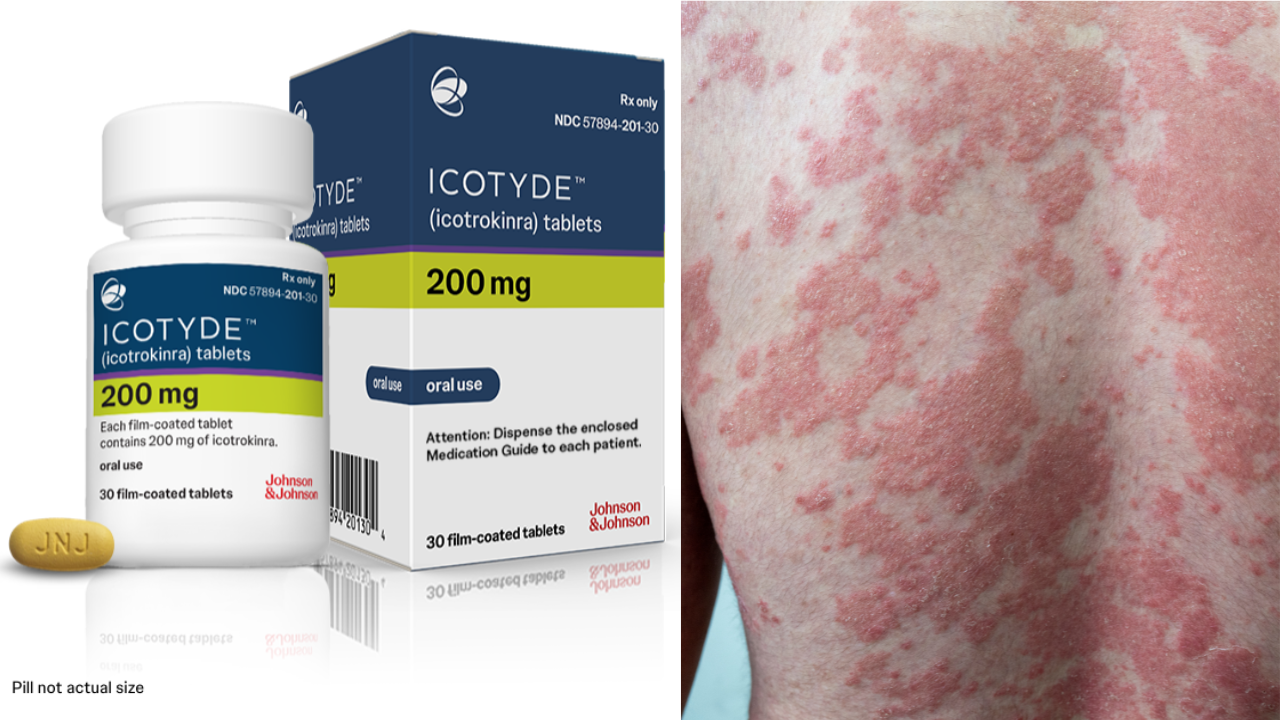
Credit: J&J/Canva
SummaryPsoriasis is an autoimmune condition that causes rough patches of skin. The new once-daily Icotyde pill, is an interleukin-23 (IL-23) receptor antagonist that has been found safe and also delivers complete skin clearance.
End of Article
