- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Addyi: FDA Approves Libido-Boosting Drug For Postmenopausal Women – What Does It Mean for Women’s Sexual Health?

Credits: Canva
U.S. health authorities have widened the approval of a closely debated medication designed to increase female libido, allowing the once-daily pill to be used by women over the age of 65. The decision, announced on Monday by the Food and Drug Administration, extends the drug’s use to older women who have already gone through menopause. The pill, Addyi, was originally cleared a decade ago for premenopausal women experiencing emotional distress linked to a low sex drive. Anisha Mathur, Founding Partner at Shepherd Law Associates, explained the development and what it means.
What Is Addyi?
Addyi, also known by its generic name flibanserin, is an FDA-approved prescription drug used to treat hypoactive sexual desire disorder, or HSDD, in women. HSDD refers to a long-term or recurring lack of sexual desire that leads to emotional distress or strain in relationships and cannot be explained by another medical condition, relationship difficulties, or medication side effects, according to the Cleveland Clinic.
Marketed by Sprout Pharmaceuticals, Addyi was once expected to become a major breakthrough in women’s health. However, the drug faced setbacks due to side effects such as dizziness and nausea. It also carries a strong safety warning about alcohol use. The FDA’s boxed warning, its strictest caution, notes that consuming alcohol while taking Addyi can lead to dangerously low blood pressure and fainting.
What Is Hypoactive Sexual Desire Disorder?
Hypoactive Sexual Desire Disorder, or HSDD, involves an ongoing reduction or absence of sexual thoughts, fantasies, and interest in sexual activity that causes significant personal distress or relationship tension. It is a common form of sexual dysfunction seen in both women and men. People with HSDD may experience low libido, little response to sexual stimulation, or a tendency to avoid sex altogether. Possible contributing factors include stress, relationship challenges, mental health conditions, certain medications, hormonal shifts, or past trauma.
The condition, which describes a distressing lack of sexual desire, has been recognised since the 1990s and is believed to affect a large number of women in the United States, based on survey data. After the commercial success of Viagra for men in the 1990s, pharmaceutical companies invested heavily in finding treatments for female sexual dysfunction. Diagnosis, however, remains complex because libido is influenced by many factors. After menopause, declining hormone levels bring biological changes and medical symptoms that further complicate assessment.
Addyi: FDA Approves Libido-Boosting Drug For Postmenopausal Women
U.S. health regulators have now expanded the approval of the controversial libido drug, allowing women over 65 to take the once-daily pill. The FDA’s announcement broadens its use to include older postmenopausal women.
According to CNN, diagnosing HSDD is challenging because sexual desire is affected by multiple physical, emotional, and psychological factors, particularly after menopause. Physicians are expected to rule out issues such as relationship stress, underlying medical conditions, depression, and other mental health disorders before prescribing the medication. The diagnosis itself remains debated, with some psychologists arguing that low sexual desire should not automatically be treated as a medical condition.
Before its approval in 2015, the FDA rejected Addyi twice, citing limited effectiveness and concerns over side effects. Its eventual approval followed a strong lobbying effort by the manufacturer and advocacy groups like Even the Score, which positioned the lack of treatment options for female libido as an issue of gender equality in healthcare.
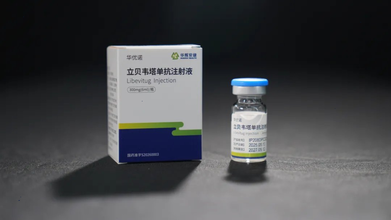
Libevitug: China Rolls Out World's First Hepatitis D Monoclonal Antibody Drug
Credit: @ChinaScience/X
China has become the world’s first country to approve a monoclonal antibody drug called Libevitug to treat hepatitis D — a severe, chronic liver disease.
The drug, which was rolled out at a Beijing hospital recently, was granted conditional approval by China's National Medical Products Administration in January 2026 following priority review, Science and Technology Daily reported.
Libevitug is a monoclonal antibody developed by a team from Tsinghua University and the Beijing-based biopharmaceutical company Huahui Health.
It works by blocking hepatitis B and D viruses from entering liver cells.
Clinical trials on Libevitug demonstrated significant efficacy in virological response and normalization of liver function, with particularly notable benefits for patients with cirrhosis.
What Is Hepatitis D? What Are The Key Symptoms?
Hepatitis D, or HDV, is different from the others—it can't infect the liver by itself. It only exists in those already infected with hepatitis B, employing the outer shell of the HBV virus to replicate.
When HBV and HDV infect the liver concurrently (a co-infection) or if HDV is contracted later on superimposition of pre-existing HBV (a superinfection), the immune system is twice assaulted. The liver has to cope with more intense inflammation, resulting in more rapid fibrosis progression and a much increased risk of cirrhosis and hepatocellular carcinoma.
In individuals with co-infection, liver injury is more severe, and complications arise years sooner than in HBV mono-infection. There are no specific therapies for HDV currently, and its management relies mostly on the control of hepatitis B.
It is also possible to prevent HBV infection and thereby hepatitis D by vaccination. Antiviral therapy and close follow-up can likewise decrease the chances of serious HDV complications in those already infected with HBV.
Simultaneous infection with HBV and HDV can lead to mild-to-severe hepatitis, with signs and symptoms typically appearing 3–7 weeks after initial infection and include:
- fever,
- fatigue,
- loss of appetite,
- nausea,
- vomiting,
- dark urine,
- pale-colored stools,
- jaundice (yellow eyes)
- fulminant hepatitis.
Also read: Hepatitis On Rise In India: What Is Causing It?
How Does Libevitug Work?
Of the over 254 million chronic hepatitis B carriers worldwide, approximately 5 percent are co-infected with the hepatitis D virus, and patients with co-infection have long lacked effective targeted therapies.
Clinical trials for the drug began in 2018, with an international multi-center study launched in 2023 among patients with chronic co-infection.
The drug previously received "Breakthrough Therapy Designation" from both the Center for Drug Evaluation (CDE) of the China NMPA and the US Food and Drug Administration (FDA).
Also read: Hepatitis A To E: How Each Virus Affects Your Liver?
"Libevitug demonstrates outstanding clinical efficacy and significantly improves liver stiffness, with particularly remarkable efficacy in patients with HBV/HDV-related cirrhosis," stated Professor Niu Junqi, principal investigator of the Libevitug trial from the First Hospital of Jilin University.
"This drug aligns with the core objectives of China's Action Plan for the Prevention and Control of Viral Hepatitis (2025–2030), which aims to improve diagnosis and treatment rates for viral hepatitis and reduce the incidence of liver cancer and mortality," Junqi added.
Junqi noted that Libevitug is expected to effectively lower the risk of disease progression to advanced severe diseases such as liver cancer, drive the diagnosis and treatment of HDV in China, as well as advance the goal of eliminating viral hepatitis as a public health problem by 2030.
India Launches New Yoga Protocols To Fight Diabetes, High BP, Asthma

Credit: Canva
Amid the growing burden of Non-Communicable Diseases (NCDs), India’s Union Ayush Ministry has launched yoga protocols that will target vulnerable groups with diabetes, high blood pressure, and asthma.
The “Yoga Protocol for Non-Communicable Diseases (NCDs) and Target Groups” is a significant push towards reshaping the country’s healthcare narrative from treatment to prevention, PTI reported.
NCDs such as diabetes, hypertension, cardiovascular ailments, chronic respiratory conditions, and mental health disorders are increasing significantly in the country. These also account for about 60 percent of all deaths in the country, signaling a clear need to focus on preventing the lifestyle-driven illnesses.
Also Read: IPL 2026: Rohit Sharma's Transformation Impresses Nita Ambani; Fitness Secrets Revealed
Prataprao Jadhav, Union Minister of State (Independent Charge) for Ayush, noted that prevention of these conditions is important for the better health of the nation, and yoga is India’s answer to the rising burden of lifestyle diseases.
“Through these evidence-based protocols, we are empowering every citizen to take charge of their own health and well-being in a simple, accessible, and sustainable manner. By integrating yoga into daily life, we aim to shift the focus from illness to wellness, reducing long-term healthcare pressures,” he said.
“This initiative reflects our commitment to building a healthier nation through holistic, preventive, and people-centric approaches rooted in India’s rich traditional knowledge,” the minister added.
What Are The Protocols?
It is part of the Yoga Mahotsav 2026, launched earlier this month by Union Ayush Minister Prataprao Jadhav, developed by the World Health Organization (WHO).
The protocols are designed as structured, evidence-based modules that integrate Yogic practices into daily life in a simple, accessible, and scalable manner.
The initiative has specific yoga interventions curated for major health conditions such as:
- diabetes — improving metabolic balance and glycemic control;
- hypertension — calming the nervous system and regulating blood pressure;
- bronchial asthma — strengthening respiratory capacity and improving lung function
- emotional well-being — breathing and meditation aimed at reducing stress, anxiety, and depression.
Further, it includes
- playful yoga modules for young children
- mental health-focused routines for adolescents,
- mobility-enhancing practices for the elderly
- specialized guidelines for women and pregnant mothers.
Yoga 365
The Ministry of Ayush has also launched Yoga 365 — a nationwide campaign to make yoga an integral part of citizens' daily lives — that goes beyond the International Day of Yoga (IDY), marked annually on June 21 worldwide.
According to the National Sample Survey (NSS), awareness of traditional systems like yoga stands at 95 percent in rural India and 96 percent in urban India.
The NSS also found that in around 1.1 crore households in rural India and around 1.4 crore households in urban India, at least one household member regularly practices yoga.
The Yoga 365 campaign aims to
- mobilize communities through mission-mode outreach,
- promote behavior change,
- integrate yoga into diverse spaces—from schools and offices to neighborhood groups and digital platforms.
The Morarji Desai National Institute of Yoga, under the Ministry of Ayush, has also signed a Memorandum of Understanding with the wellness platform Habuild to offer free daily online yoga sessions.
COVID-19 Cicada Variant: Will It Become The Dominant Strain In The US? Know All About The Virus
Credit: Canva
The COVID-19 BA.3.2 strain, nicknamed Cicada Variant, is spreading rapidly in the US, raising the possibility of becoming the dominant strain in the country, according to experts.
The US Centers for Disease Control and Prevention (CDC), in its latest report, has raised concerns about BA.3.2 — a highly mutated variant of COVID.
BA.3.2 has already been reported in at least 23 countries, including 25 states in America, as per the report. It has also been detected in 132 wastewater samples from Massachusetts.
Also Read: Bipolar Disorder: How Early Detection Can Help Prevent Serious Complications
The 25 states include California, Connecticut, Florida, Hawaii, Idaho, Illinois, Louisiana, Maine, Michigan, Maryland, Massachusetts, Missouri, New Hampshire, New Jersey, Nevada, New York, Ohio, Pennsylvania, Rhode Island, South Carolina, Texas, Utah, Vermont, Virginia, and Wyoming.
"Cicada can become the dominant strain in the US,” Dr. Robert H. Hopkins Jr., medical director of the National Foundation for Infectious Diseases, told USA TODAY.
While it is not certain, it can also "drive a US summer surge.”
The CDC warned that a new variant "with substantial capacity to evade immunity from previous infections or vaccines could be associated with seasonal increases in COVID-19 activity.”
In the US, COVID-19 is still linked to roughly 300 to 500 deaths per week, according to recent data based on provisional estimates from the CDC.
Brandon Dionne, an associate clinical professor of pharmacy and health systems sciences at Northeastern University, stated that the virus may evolve and increase the possibility of more severe disease.
“It’s something we definitely want to monitor,” Dionne said. “It could, over time, become the dominant strain in the US, but it’s not yet there," he said.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
What Is The Cicada COVID Variant?
Cicada was first identified in a respiratory sample in South Africa in November 2024.
It is a descendant of the Omicron BA.3 lineage, and is genetically distinct from the previously circulating JN.1 lineages (including LP.8.1 and XFG).
BA.3.2 comprises two major branches, BA.3.2.1 and BA.3.2.2. BA.3.2.2 also has substitutions like: K356T, A575S, R681H, and R1162P.
The World Health Organization (WHO) has designated BA.3.2 as a Variant Under Monitoring (VUM). It means the variant may not be that dangerous yet, but it may have concerning mutations.
The CDC’s latest Morbidity and Mortality Weekly Report explains that Cicada has “70 to 75 substitutions and deletions in the gene sequence of its spike protein”.
Also read: Maternal Vaccination During Pregnancy Can Prevent COVID-related Hospitalization In Babies: Study
Will Current COVID Vaccines Work?
The variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
“The number of mutations from JN.1 viruses makes it less likely that the current vaccines will be highly effective against Cicada, but we need more data to better answer this question,” Dr. Hopkins Jr. said.
However, according to the WHO, current COVID vaccines are expected to continue protecting against severe disease. Moreover, the WHO said BA.3.2 doesn’t seem to be making people sicker so far and hasn’t resulted in increased hospitalizations and deaths.
Common symptoms of Cicada variant include:
- Cough
- Fever or chills
- Sore throat
- Congestion
- Shortness of breath
- Loss of smell or taste
- Fatigue
- Headache.
© 2024 Bennett, Coleman & Company Limited

