- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
FDA Has Approved A New Medication Treatment For Migraines In Adults

(Credit-Canva)
Headaches are a common symptom of a stressful lifestyle, your body not feeling well and other issues. While headaches can be dealt with easily, migraines are not so easy to handle. Migraines are a type of headache that feels like severe throbbing and pulsing sensation, almost like you are hearing your own heartbeat in your brain, usually on one side of the brain. Many people believe that migraines are not that big of a deal because you just have to deal with the pain, but that is not all migraine is, some people find it very difficult to do their daily tasks as they experience dizzying spells, nausea and extreme sensitivity to light and sound! These attacks can last hours and make it difficult for people to go about their daily lives as well. While there are medications available for migraine patients, these medications need time to take effect, so you may be in a lot of pain, but there are not many quick reliefs you can have in place other than learning the symptoms of a migraine attack and taking medication before it happens. But a new approval by the FDA may change this!
The U.S. Food and Drug Administration (FDA) has approved Symbravo, a new medicine to treat acute migraine attacks in adults. This means adults can now use Symbravo to get relief from their migraine symptoms. The FDA's decision was based on the results of three big studies, called Phase 3 trials. These trials involved over 21,000 migraine attacks, so the FDA has a lot of information about how well Symbravo works and how safe it is. The FDA only approves medicines that have been shown to be both safe and effective through a thorough testing process.
Importance of New Treatment Option
"Migraine attacks can happen suddenly and really mess up people's lives. It's estimated that over 39 million people in the U.S. alone get migraines," said Herriot Tabuteau, M.D., CEO of Axsome Therapeutics told US News. This shows how common migraines are and how important it is to have good treatments. "Symbravo gives patients and doctors a new option that can quickly stop a migraine attack, keep it away, and let people get back to their normal activities, all with just one dose." Having a medicine that can give fast and long-lasting relief from migraine pain is a big deal for millions of people. This new treatment is a real step forward in how we treat migraines.
Symbravo Trial Results, How Was It Approved
The trials took place in 3 steps, the Momentum trial study focused on people whose migraines had moderate to severe pain. The results showed that a lot more people taking Symbravo felt pain-free two hours after taking the medicine compared to those who took a placebo which is a dummy pill. Even better, many people felt relief for up to 24 and even 48 hours after just one dose. This long-lasting relief is really important for people with migraines because it means they can get back to their normal lives without worrying about the pain coming back. The study also looked at how many people were free from their worst symptom, like sensitivity to light or sound, or nausea. Symbravo worked better than the placebo in this area too.
While the intercept trial looked at people who took Symbravo when their migraine pain was still mild. Even when the pain was just starting, Symbravo was effective. The results were similar to the MOMENTUM trial, with many people getting pain relief and relief from their worst symptoms. Treating migraines early is often better because it can stop the pain from getting really bad.
And lastly the Movement trial which was to see how safe the medication is when people take it regularly. This study followed 706 people who had at least two migraines a month. The most common side effects people experienced were sleepiness and dizziness. While these side effects are important to know about, the study showed that Symbravo is generally safe for people to use on a regular basis.
Top Indian Medical Body Declares Stem Cell Therapy For Autism Illegal

After a recent Supreme Court of India order banning the use of stem cell therapy to treat autism -- a neurodevelopmental condition affecting communication, social interaction, and behavior -- the country's National Medical Commission has issued a clear warning to doctors and hospitals not to use the therapy to treat autism in routine medical practice.
The National Medical Commission, in a new advisory, asked all medical colleges, hospitals, and doctors to strictly follow the rules.
In a letter sent to the regulator, Indian Council of Medical Research Director-General Dr. Rajiv Bahl stated that the stem cell treatment can now be used in regular medical practice only for 32 diseases that are officially approved by the government. These include blood cancers and serious blood disorders such as:
Acute myeloid leukemia
Thalassemia
Multiple myeloma
Aplastic anemia
Myelofibrosis
Germ cell tumors.
The letter asked doctors not to offer stem cell therapy for any other disease outside this list.
Notably, the Ethics and Medical Registration Board (EMRB), under the National Medical Commission (NMC), had, in December 2022, constituted the Committee on Stem Cell Use in Autism Spectrum Disorder (ASD).
It had stated that none of the current international guidelines recommend stem cell therapy as a treatment for ASD and added that the therapy is not recommended as a treatment for ASD in clinical practice.
What Was The Supreme Court Ruling On Stem Cells?
Earlier this year, in January, a bench comprising Justice JB Pardiwala and Justice R Mahadevan noted that stem cell therapy lacks “scientific support and has not been recognized as a sound medical practice backed by empirical evidence”.
The Bench ruled that “every use of stem cells in patients outside an approved clinical trial is unethical and shall be considered as malpractice.”
The apex Court, however, added that the advanced therapy, which holds promise in several medical fields, can still be approved for monitored clinical research trials. It added that the patients have the liberty to participate in approved and regulated clinical trials.
What Is Stem Cell Therapy
Stem cell therapy, also called regenerative medicine, is a medical treatment that uses stem cells to repair or replace damaged tissues.
While the therapy is useful and effective for blood cancers and autoimmune diseases, for the treatment of autism, there is no proof or scientific evidence of its utility.
As stem cell therapy is vastly unregulated in India, many private labs have been minting money over the promise of treatment for autism.
“Most stem cell therapies are unregulated in India and are promoted based on no evidence, and fake advertisements. While these disorders have no cure, many people are falsely lured by these companies,” Dr. Manjari Tripathi, Head of Department, Neurology, AIIMS Delhi, had told IANS, after the SC verdict.
Stem Cells Permissible Only For Research
The NMC warned that any doctor or institution offering stem cell therapy for autism will face regulatory and legal action.
The top medical regulator, however, stated that stem cell therapy is permissible only for research purposes.
Such studies must follow strict rules set by the government and must be approved by ethics committees and national regulatory bodies.
Researchers must also ensure that patients give written consent, that treatment is provided free during the trial, and compensation is offered if injury or death occurs during research.
Florida Restores H.I.V. Drug Access With 31 Million Emergency Funding

Credits: Canva and Wikimedia Commons
Florida on Tuesday approved nearly $31 million in short-term funding for H.I.V. medication for residents This is a reverse course after the state health officials restricted the access to a program that helps only those who could afford costly medications.
What Happened In The Past?
The Florida Department of Health had imposed tougher eligibility rules on March 1 that left 12,000 residents with H.I.V. without the access to medication covered by the state's AIDS Drug Assistance Program. This deprived them of life saving drugs, noted a nonprofit advocacy group - AIDS Healthcare Foundation.
The foundation also sued the department over these changes with protestors rallying in opposition at the State Capitol in Tallahassee. The department cited a projected $120 million state budget that restricted eligibility for the program.
Read: HIV vs. AIDS: What You Should Know About These Commonly Confused Terms
Yet, the Florida House of Representatives and State Senate, both led by Republicans, unanimously approved legislation that provides $30.9 million to fund the program through June 30 and restore the eligibility rules. Gov. Ron DeSantis, a Republican, signed the bill into law on Tuesday.
“Lawmakers on both sides understood the urgency of the crisis,” State Senator Carlos Guillermo Smith, a Democrat from Orlando who spoke out in support of the bill on the floor, said in an interview on Wednesday. “It is a matter of life and death. People had already lost access to their medications.”
Restriction On AIDS Drug Assistance Programs
As per an analysis by KFF - a health research group, Florida was one of the nearly 20 states, led both by Republicans and Democrats that imposed restrictions on AIDS Drug Assistance Programs.
The programs cover the cost of H.I.V. medications, which can run into thousands of dollars each month. They provide drugs free to some individuals and help others by paying their insurance premiums. Overall, they support about a quarter of the 1.2 million people living with H.I.V. in the United States.
However, these programs are under growing pressure. The cost of H.I.V. medications continues to rise, and the expiration of health care subsidies has led to a sharp increase in insurance premiums. Despite this, federal funding for these programs has remained largely unchanged for more than a decade.
In Florida, the Department of Health had reduced eligibility for the state’s AIDS Drug Assistance Program to 130 percent of the federal poverty level, which is about $20,748 a year for an individual, down from 400 percent or $63,840. A new law has now reversed this change, restoring eligibility back to the 400 percent threshold.
“For 10 weeks, 12,000 Floridians living with H.I.V. did not know if they could fill their next prescription,” Esteban Wood, the director of advocacy and legislative affairs at the AIDS Healthcare Foundation, said in a statement on Tuesday. “Today, they can.”
US FDA Approves Drug To Treat Rare Childhood Syndrome
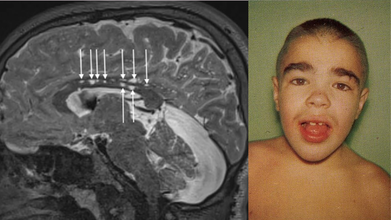
Credit: Wikimedia Commons
The US Food and Drug Administration (FDA) has approved a drug therapy to address neurologic symptoms of a rare genetic disorder — Hunter syndrome.
The X-chromosome-linked disease, occurring predominantly in males, has an estimated 2,000 affected individuals worldwide.
The drug Avlayah, developed by pharma company Denali Therapeutics, targets certain individuals with Hunter syndrome (Mucopolysaccharidosis type II or MPS II).
It is the first therapy to address the neurologic complications of Hunter Syndrome, such as
- progressive cognitive decline,
- behavioral problems,
- hydrocephalus,
- spinal cord compression,
- seizures,
- carpal tunnel syndrome.
“Today is a milestone day for children and their families battling Hunter syndrome,” said FDA Commissioner Marty Makary, in a statement.
“Avlayah is the first product approved to address neurologic complications of Hunter Syndrome, a very rare and often severe X-linked disorder in children, affecting about 500 people in the US, almost exclusively males,” added Acting CDER Director Dr. Tracy Beth Hoeg.
The FDA noted that Avlayah, the once-weekly drug given via IV infusion, must begin in presymptomatic or symptomatic pediatric patients weighing at least 5 kg before advanced neurologic impairment.
What Is Hunter Syndrome?
Hunter syndrome is a rare inherited lysosomal disorder in which sugar molecules called glycosaminoglycans build up within the cells’ lysosomes.
The condition affects physical and mental development and causes abnormalities in the skeleton, heart, respiratory system, brain, and other organs.
Hunter syndrome is a rare congenital metabolic disease. It was first reported in 1917 by a Canadian physician, Charles Hunter, in two brothers in a family.
The brothers presented typical signs, such as
- short stature,
- inguinal hernia,
- macroglossia,
- enlarged skull,
- decreased hearing,
- coarse facial features,
- protruded abdomen with hepatosplenomegaly,
- umbilical hernia,
- skeletal deformities.
The younger brother had symptoms of Central Nervous System (CNS), including seizures and cognitive decline, while the older brother did not have CNS involvement.
The estimated incidence is 1 in 162,000 live male births.
How The FDA Approved Avlayah
The FDA approval came after Avlayah showed promise in reducing cerebrospinal fluid heparan sulfate — one of the glycosaminoglycans that accumulates in the body in this disorder and is linked to the organ damage that occurs in early childhood.
The phase 1/2 multi-cohort, single-arm, open-label trial enrolled 47 pediatric patients with Hunter syndrome aged 3 months to 13 years.
Of these, 44 patients with measurements at had a 91 percent average decrease from baseline in CSF.
Denali is now conducting a randomized clinical trial that is more than 95 percent enrolled to evaluate the clinical benefit of this product.
"In the meantime, families with young children with Hunter Syndrome will have access to a product that may favorably alter the course of the disease at the crucial time in life when there is the greatest potential for benefit," Hoeg said.
Are There Any Side Effects?
Avlayah’s labeling includes a boxed warning for allergic reactions, including anaphylaxis, associated with the drug.
The FDA recommended that patients start therapy in a health care setting with appropriate medical monitoring and support measures.
The common side effects of Avlayah include
- upper respiratory tract infection,
- ear infection,
- fever,
- anemia,
- cough,
- vomiting,
- diarrhea,
- rash,
- COVID-19,
- runny nose,
- nasal congestion,
- fall,
- headache,
- skin abrasion,
- hives.
The FDA also suggested that healthcare workers monitor
- hemoglobin levels due to the risk of anemia,
- kidney function and urine protein levels due to
- the risk of membranous nephropathy (a kidney disease).
© 2024 Bennett, Coleman & Company Limited

