- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Indian Drug Regulator Flagged 90 Combination Medicines Sold Without Approval

Credits: Canva, iStock and cdsco.gov.in
Indian drug regulator - Central Drugs Standard Control Organisation (CDSCO) found 90 combination medicines that are sold without mandatory central approval. This has raised concerns on regulatory compliance in the country's pharmaceutical supply chain.
CDSCO is the apex drug regulatory body in the country. The authority wrote a letter to all states and union territory drug controllers and asked them to investigate manufacturers and marketers of these fixed-dose combinations (FDCs).
What are FDCS? These are drugs that contain two or more active ingredients in a single formulation. The CDSCO asked them to investigate and submit reports on the action taken at the earliest.
What Made To The Unapproved List Of FDCs?
The annexure attached to the letter showed 90 combination medicines had made it to the market. These contain cough syrups, vitamins, and anti-diabetic pills.
Cough And Cold Medicines
Cough and cold combination medicines have formed the single largest group. At least 14 to 16 entries show combination of ingredients found in everyday pharmacy shelves: dextromethorphan, ambroxol, guaiphenesin, chlorpheniramine, phenylephrine, terbutaline, and menthol in various permutations. Several of these are sold as flavoured syrups for children. Budesonide-levosalbutamol inhalation suspension that is used for asthma and chronic obstructive pulmonary disease, too features in the list.
Nutritional Supplements
The next group is of the vitamins, minerals and other nutritional supplement which has roughly 18 to 20 entries. They include:
- Multivitamin capsules
- Iron-folic acid syrups
- Calcium-vitamin D3-K2 combinations
- Abapentin-methylcobalamin preparation
Antifungal and Dermatological Combinations
This group has accounted for at least 10 entries, which include antifungal creams that could lead to potential misuse and skin damage when sold without prescriptions.
Anti-diabetic Medicines
This has accounted for six entries, and the combination includes:
- Glimepiride paired with metformin or pioglitazone
What Did CDSCO Write To Drug Controllers?
The letter dated 18 March was signed with Drugs Controller General of India (DCGI), Dr Rajeev Singh Raghuvanshi. He described the matter as being "top priority in the interest of public health". The unapproved drugs were detected during a scrutiny of laboratory testing data submitted through SUGAM, which is CDSCO's online portal for drug sample testing, for the year 2025.
“This is to inform you that upon scrutiny of the SUGAM lab testing data for the year 2025, a large no. of drug samples (FDCs) are detected as unapproved and fall under the category of ‘New Drug. No new drug shall be manufactured for sale unless it is approved by the licensing authority."
What Is India's Rule For FDCs?
Under India's New Drugs and Clinical Trial Rules of 2019, any FDC is treated as a new drug and must have the central government's approval before it could be manufactured and sold. The regulator's letter noted that there were presence of unapproved drugs in the supply chain that could pose risks to public health. This also amounts to a violation of the Drugs and Cosmetics Act, 1940.
“The presence of unapproved drugs in the supply chain is a matter of serious concern, posing potential risks to public health and safety. It also indicates non- compliance with the provisions of the Drugs and Cosmetics Act, 1940, and Rules made thereunder," it said.
DCGI urged states and UTs "to examine the unapproved FDCs in generic name as mentioned in the attached annexure". This is done to check whether any approval has been granted by the local office. "Accordingly, please initiate appropriate investigation and regulatory action against the concerned manufacturers, marketers, and other stakeholders, as deemed fit. Kindly ensure strict monitoring and enforcement to prevent the manufacture, sale, and distribution of such unapproved drugs," the letter noted.
Royal Gwent Hospital Treated 21 People With Unsterilized Surgical Instruments, Patients May Be Infected

Credits: Google Photos
Royal Gwent Hospital in Newport, Wales treated 21 patients with unsterilized surgical instruments. The patients were not informed of the error for three weeks. As per a BBC report, the instruments were disinfected but not medically sterilized, which is a typical medical protocol. On February 24, the outlet carried out a breach of protocol, and then on February 25 and 26, the same instruments were used in operations and examinations.
While the error was discovered by hospital staff on February 27, the patients were not informed till March 16.
Also Read: Indian Drug Regulator Flagged 90 Combination Medicines Sold Without Approval
What Happens When Unsterilized Hospital Instruments Are Used?
Surgical tools that are improperly sterilized could spread bloodborne diseases like HIV and hepatitis. In a statement obtained by the BBC from Aneurin Bevan University Health Board, the organization that manages Royal Gwent Hospital, a spokesperson said the situation is being investigated. The spokesperson also said that they have arranged for "precautionary testing" for all affected patients, and that "the clinical risk of bloodborne virus exposure is extremely low".
"We fully recognize the concern and distress this may cause, and we are truly sorry. The well-being of our patients is our highest priority, and we are taking all necessary actions to understand how this happened and to prevent it from occurring again,” the board's statement continued.
They additionally stated that “this was a very limited incident” and that “there is no wider cause for concern.”
Family Speaks Out After Teen Patient Affected

One of the patients affected is 15-year-old Ieuan, who underwent an orthodontic procedure at Royal Gwent Hospital. His parents said the call from the hospital informing them about the sterilization error came as a shock.
According to the family, the news was difficult to process, especially because their son will now have to undergo a series of medical tests over the next six months. Ieuan later said he initially felt frightened after learning about the possible exposure and is hoping the tests confirm he is safe.
The teenager has already undergone his first blood test at the hospital and is scheduled for additional tests in the coming months before doctors can give him a final all clear.
What Went Wrong During The Procedure
Ieuan had braces fitted on February 25 during a routine orthodontic appointment to address gaps in his teeth following an earlier dental operation.
During the procedure, staff used instruments including pliers used to trim brace wires and a dental mirror. The hospital later confirmed that while the tools had been disinfected, they were not placed in an autoclave, the specialized machine that sterilizes medical equipment using high heat and steam.
Standard protocol requires staff to confirm that sterilization indicators on instrument trays have changed color after the process and to check the sterilization date before use. According to the health board, this verification step was missed due to what it described as human error.
Health officials say investigations are ongoing, and monitoring of affected patients will continue over the coming months.
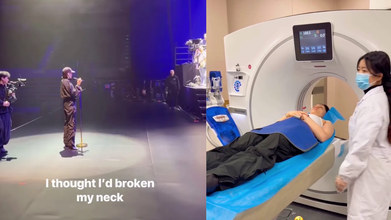
Jessie J Undergoes MRI Scan After Car Injury, Feared She Broke Her Neck
Credits: Instagram
Jessie J, 37, was rushed to hospital after a car injury that left her in fears that she broke her neck. She underwent an MRI scan after the incident and kept her fans on loop through her Instagram update.
Currently in China, the singer is there for her No Secrets Tour. She said that she suffered an injury after hitting her head on the roof of the car. The singer had shared a video where she was climbing into the back of the car after her latest concert and said she "didn't mind squashing in the back". The video then cuts to her being in the hospital undergoing an MRI scan after she had suffered a neck injury.
She said, "Yeah, I just can't move my head. I'm alright. I'll just have to take some painkillers and march right through it." She further said, "I thought I'd broken my neck, but I haven't. But I have really hurt my neck and my back."
In her later shots, she was seen trying on her gold jumpsuit while embracing her young son Sky. However, she did not let this injury stop her from performing. Many fans also commented her messages of love and well wishes.
Jessie J Health Update
Previously, the singer took to her Instagram to share her stage-one breast cancer diagnosis, fans everywhere were left both shocked and motivated. The 37-year-old singer-songwriter, announced the news in typical frankness, mixing vulnerability with as she explained the surgery she'd be having after performing at Capital's Summertime Ball at London's Wembley Stadium.
In her heartfelt video message, Jessie shared that she has been “in and out of tests” recently and ultimately received a diagnosis of early breast cancer. “Cancer sucks in any form,” she said, “but I’m holding on to the word early.”
With a nod of dark humor, she added, "It's a very dramatic way to get a boob job," vowing followers, "I will come back with massive boobs and more music." But under the humor lay raw emotion and an earnest appeal for sympathy and understanding. "I need to process it and talk about it," she said. "I need a hug."
Having struggled with a string of serious health problems in the past, including a childhood heart condition, a stroke at age 18, Meniere's disease, and even recently being diagnosed with ADHD and OCD, Jessie J is certainly no newcomer to working through health problems. But this latest installment has introduced her to an even more intimate, more personal view.
By disclosing her diagnosis, Jessie J is part of a rising number of public figures using their influence to make health screening and breast cancer discussions more mainstream. Such openness is particularly important in light of the fact that many young women still unrealistically believe breast cancer is an "older woman's disease."
Alkem Laboratories Launches Cheapest Semaglutide Injection In India
Credit: Canva
India's Alkem Laboratories is all set to launch a semaglutide injection, meant for weight-loss and diabetes patients, priced merely at weekly cost of Rs 450 - making it one of the cheapest options available in the market.
The patent for Novo Nordisk, the original maker of semaglutide, ended on March 20 and since then, numerous pharmaceutical companies have begun releasing their own versions of the blockbuster drug.
Alkem Laboratories's drug, marketed under the brand names Semasize, Obesema and Hepaglide in India, will is administered as a once-weekly subcutaneous injection and the organization has revealed a pre-filled disposable injection pen priced at Rs 1,800 for a month’s dosage.
Apart from the drug, Alkem is also offering a reusable injection pen for higher maintenance doses, allowing patients to replace only the medication cartridge instead of buying a new device each time in order to reduce long-term treatment costs and improving adherence.
The company has received approval from the Drug Controller General of India to manufacture and market semaglutide for Type 2 Diabetes Mellitus and chronic weight management, following a review of Phase 3 clinical trials conducted in India.
Other Semaglutide Injection Launches In India
Apart from this, NATCO Pharma became the first to introduce Semanat and Semafull for INR 1,290 per month and INR 1,750, about 90 per cent cheaper than Ozempic, costing INR 8,800 per month.
Eris Lifesciences also announced its plans to launch a multi-dose vial under the brand name Sundae at the same price. Dr. Reddy’s Laboratories, Sun Pharmaceutical Industries, and Glenmark Pharmaceuticals launched their generic versions of semaglutide. Dr. Reddy’s Obeda is priced at Rs 4,200 per month, available in 2 mg and 4 mg strengths.
Sun Pharmaceutical Industries will sell under the brand names Noveltreat (doses ranging from INR 900) and Sematrinity (doses ranging from INR 750). Meanwhile, Glenmark’s GLIPIQ vials range from Rs 325 to Rs 440 per week.
Zydus Lifesciences has also announced the launch of its generic version of semaglutide injection under the brand names SEMAGLYNTM, MASHEMATM, and ALTERMET. The average monthly cost of the treatment will be about Rs 2,200, the company said in a statement.
Semaglutide: Risks Vs Benefits
In people with type 2 diabetes, Harvard Health noted that the body's cells are resistant to the effects of insulin and the body does not produce enough insulin, or both. This is when GLP-1 agonists stimulate the pancreas to release insulin and suppress the release of another hormone called glucagon.
These drugs also act in the brain to reduce hunger and act on the stomach to delay emptying, so you feel full for a longer time. These effects can lead to weight loss, which can be an important part of managing diabetes.
Notably, semaglutide is not just a glucose-lowering drug, it improves weight and has proven cardiovascular, liver, and renal benefits.
Wider availability could therefore help reduce long-term complications such as heart disease and kidney failure. If used appropriately, this could indeed be a major step forward in total metabolic care, the experts said.
“The increasing affordability of GLP-1 receptor agonists is a double-edged development. On one hand, it significantly improves access for patients with obesity, type 2 diabetes, and high cardiovascular risk, conditions that are highly prevalent in India. These drugs have demonstrated meaningful benefits in weight reduction, glycemic control, and even cardiovascular risk reduction, which could translate into long-term public health gains,” Dr Vivek Bindal, Senior Director & Head, Bariatric and Robotic Surgery, at Max Healthcare, told HealthandMe.
A recent study published in The Lancet Psychiatry journal showed that Ozempic can also help tackle the burden of depression, anxiety, and self-harm in high-risk diabetic patients.
However, the benefits are conditional as it depends on appropriate prescribing, reliable product quality, and adequate patient monitoring, said Dr. Anoop Misra, who heads Delhi’s Fortis-C-DOC Centre for Diabetes and Endocrinology, told HealthandMe..
"These drugs should be strictly prescribed by qualified doctors and used only for therapeutic purposes, not for cosmetic weight loss,” Dr. Mohan said.
Urging for careful monitoring of side effects, the expert also called for more Indian data and strict pharmacovigilance.
“This also presents a great opportunity for India to make these drugs affordable and accessible to other developing countries across Asia and Africa,” the noted diabetologist said.
The experts also warned of “indiscriminate or unsupervised use” due to low cost.
This is particularly for cosmetic weight loss without proper medical evaluation, said Dr. Bindal.
“GLP-1 therapies are not ‘quick fixes’ -- they require careful patient selection, dose titration, and monitoring for side effects such as gastrointestinal intolerance or, rarely, pancreatitis,” he added.
However, the focus should remain on holistic obesity management, including lifestyle modification, rather than over-reliance on pharmacotherapy alone.
© 2024 Bennett, Coleman & Company Limited

