- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
Popular Sleep Aid Suspended After Gummies Triggered Kids 'Overdosing' On Melatonin

Credits: Canva
A major online health retailer has suspended sales of melatonin products to Australia after a surge in overdose reports, raising alarm about the widespread and often unregulated use of the hormone as a sleep aid for children.
US-founded iHerb, which delivers supplements and health products to more than 180 countries, abruptly pulled melatonin from its Australian marketplace. The move has left many parents in shock, particularly those who have long relied on the company to sidestep prescription-only rules in Australia.
For years, families have quietly ordered melatonin from overseas to help children with ADHD, autism spectrum disorder, or chronic sleep problems. Many never consulted a doctor, trusting online reviews or anecdotal reassurance instead.
Melatonin Gummies: Parents Divided Between Panic and Relief
For some families, iHerb’s decision feels like a lifeline suddenly cut off. One mother, posting in a support group for parents of neurodivergent children, admitted: “I know a lot of us purchase melatonin from iHerb for our ADHD/ASD kiddos. It’s now been banned due to it being a prescription medicine.”
Another confessed the impact was immediate: “My kids won’t sleep without it.”
But the suspension has also sparked relief among others who fear melatonin has become dangerously normalized. One parent commented, “Honestly, I think this is a good thing. My son is autistic and I use it for him so I understand some children really do need it – but it’s also very overused, and look at all the overdoses from it this year.”
This divide reflects a broader tension in child health: the desperate search for effective solutions versus the risks of self-managing with little medical oversight.
A Surge in Poisonings Raises Red Flags
The catalyst for iHerb’s decision appears to be stark new data out of Western Australia. Calls to the state’s Poisons Information Centre about melatonin overdoses have jumped 166 percent in the past five years, with nearly 90 percent involving children.
In 2018, the hotline logged 175 calls. By August this year, that number had soared to 322.
The majority of cases involve children accidentally consuming too many melatonin gummies. Symptoms reported included stomach pain, dizziness, excessive drowsiness, and headaches. Though rarely fatal, repeated overdoses underscore how the supplement’s candy-like form and lack of dosage regulation put children at risk.
The rise of melatonin gummies has reshaped how parents approach sleep issues. Marketed as natural, gentle aids, they blur the boundary between dietary supplement and pharmaceutical.
Some products even fail to disclose melatonin on their packaging, presenting them instead as magnesium or herbal-based sleep support. Investigations have revealed discrepancies in labeling, with some gummies containing significant levels of melatonin despite being marketed differently.
This grey zone is particularly troubling because, in Australia, melatonin is legally restricted. It is classified as a Schedule 4 prescription-only medicine, available to adults over 55 for short-term insomnia or to travelers for jet lag. For children, it may be prescribed in specific clinical cases, but under strict medical guidance. The online marketplace bypassed all of that until now.
A spokesperson for iHerb told the Daily Mail, “The health and well-being of our customers always comes first. That’s why we are suspending all melatonin sales to Australian customers while we further review our order protocols for these products. Our goal is to support wellness responsibly and to give our customers confidence that they can shop with safety and trust.”
The company has not provided a timeline for when or if sales might resume. Dr. Kyle Hoath of the Australian Medical Association’s Western Australian branch welcomed the suspension. “It’s good to see that iHerb has taken a proactive step,” he said. “The problem is that, when you think about the way an online retailer like iHerb works, there’s no interaction with a pharmacist. It’s the consumer choosing what they want without any guidance around what is safe.”
The move highlights how online platforms can fill regulatory gaps but also create new ones, enabling mass importation of products that would otherwise face strict scrutiny.
While Australia is grappling with the risks of imported melatonin, other countries face similar challenges. In the United States, melatonin is widely available as an over-the-counter supplement, with minimal regulation compared to pharmaceuticals. Usage has skyrocketed among children and teenagers, prompting pediatricians to raise alarms about potential long-term impacts on hormone development and circadian rhythms.
In the UK, melatonin is also tightly controlled, available only by prescription. Yet similar concerns about misleading labeling and accidental overdoses in children have emerged there too. This patchwork of rules creates opportunities for cross-border loopholes—like Australian parents ordering through iHerb that complicate public health responses.
For parents who depended on melatonin, the suspension may feel devastating. But health experts stress that the bigger issue isn’t the lack of access—it’s the lack of medical oversight.
Children’s sleep problems can stem from many causes: neurological conditions, behavioral habits, environmental triggers, or emotional stressors. While melatonin may help in certain cases, it is not a one-size-fits-all solution. Without guidance from a pediatrician, families risk masking deeper issues or creating new ones.
The iHerb suspension is not just a supply chain hiccup it’s a wake-up call about the need for safe, evidence-based care.
How Can Parents Build A Better Bedtime Sleep Routine Without Dependence on Melatonin?
Parents anxious about losing melatonin as an option can take heart: natural, structured approaches to sleep often prove more effective long-term than supplements.
Creating a reliable bedtime routine is one of the most powerful tools. Children thrive on predictability, and a consistent wind-down ritual teaches the brain when it’s time to rest. This can start with dimming lights an hour before bedtime, turning off screens, and engaging in calming activities such as reading together, listening to soft music, or practicing gentle breathing.
Sleep hygiene also extends to the environment. A cool, dark, quiet room signals to the body that it’s time to sleep. White noise machines or soft nightlights can help reduce anxiety, especially for children who struggle with sensory sensitivities.
Parents should pay attention to diet and activity as well. Limiting caffeine and sugar in the afternoon, ensuring children get plenty of daylight exposure and exercise, and timing dinner a few hours before bed can significantly improve sleep readiness.
Perhaps most importantly, bedtime should not feel like a punishment. When framed as a positive, nurturing routine—one that involves connection rather than control children are more likely to settle into it willingly.
While melatonin may play a role for some children under medical supervision, it cannot replace the emotional security and consistency that a healthy routine provides. For families suddenly cut off from supplements, this may be the right moment to reset and rebuild sleep in a sustainable, safe way.
New Zealand Launches Surveillance After Spotting Dengue, Zika-carrying Mosquito Larvae

Credit: Canva
New Zealand has stepped up surveillance after the first detection of a dengue and Zika-carrying mosquito larvae in the country.
The larval species was confirmed as 'Aedes aegypti' — known to carry diseases including dengue fever, yellow fever, Zika, and chikungunya, across the globe.
As New Zealand does not normally have the mosquito species, the larvae detected were counted as "exotic".
The mosquito species did not cause any outbreak, but were spotted during a routine surveillance program in Auckland.
"The National Public Health Service has commenced a heightened surveillance and interception programme following mosquito larvae being collected from a routine surveillance trap at Queens Wharf, Auckland, on Monday 30 March," Health New Zealand said in a statement.
Health New Zealand reported that exotic species were occasionally found at ports and airports.
Not A Public Threat Yet
The health body noted that the larvae were not considered a public health or biosecurity threat yet because there was no indication they had become established.
But the agency aimed to continue "intensive monitoring for at least three weeks".
"The monitoring would take place within a 400m radius of the site where the larvae were identified. Health Protection Officers would place mosquito traps in the survey area," the statement said.
"These have been hidden away from plain sight so they are not disturbed, for example, in old tyres, bushes, or pools of water. We ask members of the public to avoid touching or disturbing these traps if they find them, as it may disrupt our monitoring and trapping efforts," medical officer of health Dr David Sinclair said.
Sinclair said New Zealanders were most at risk from diseases transmitted by mosquitoes when travelling overseas, including to Pacific Island countries and territories where dengue fever was known to be present.
Dengue Surging In 17 Countries
The US Centers for Disease Control and Prevention recently issued a travel alert of dengue outbreaks across 17 countries.
The CDC alert issued on March 23 identified 17 countries reporting an increased number of cases of dengue. These include: Afghanistan, Bangladesh, Bolivia, Colombia, Cook Islands, Cuba, Guyana, Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, Vietnam, and the United States territories of American Samoa, Puerto Rico, and the US Virgin Islands, where local transmission is already common.
Also read: Long-term Exposure To Air Pollution Increases Fatality Rates In Dengue: Study
What Is Dengue?
Dengue is a disease caused by a virus spread through mosquito bites. It is transmitted through infected mosquitoes, primarily the species Aedes aegypti.
The breakbone fever is caused by an infection with any of four different dengue viruses. These include:
• Dengue virus type 1 (DENV-1 or DEN-1)
• Dengue virus type 2 (DENV-2 or DEN-2)
• Dengue virus type 3 (DENV-3 or DEN-3)
• Dengue virus type 4 (DENV-4 or DEN-4)
Also read: New dengue vaccine over 80% effective, prevents severe disease for up to 5 years
Common Symptoms of dengue include:
• Sudden onset of high-grade fever.
• Intense headache
• Severe muscle, joint, or bone pain.
• Skin Rash that often appears 2–5 days after the fever starts
• Nausea and Vomiting
• Minor bleeding
• Fatigue.
COVID Cicada variant: Are Children More At Risk? How To Safeguard?

Credit: iStock
The COVID Cicada variant, currently circulating in 23 countries, is likely to affect children more than the previous variants.
Children have largely escaped severe illness from the SARS-COV-2 virus.
However, the new Cicada variant with around 75 genetic changes in its spike protein is likely to disproportionately affect children, as per an expert, who noted its presence in the UK.
“Some people have done analysis on this, suggesting it may be more prevalent among young children. Children get infections all the time, but this might be something to do with the fact that they have never been exposed to Covid vaccines," Prof Ravindra Gupta, of Cambridge University, who advised the UK government during the pandemic, was quoted as saying to The Mirror.
“So this is something we’re looking at in the lab to try and work out why. The problem with this is that it is an infection that spreads fast. Eventually, it ends up in someone vulnerable," he added.
Key Symptoms
Symptoms seem to be similar to those of other recent variants and include
- sore throat,
- cough,
- congestion,
- fatigue,
- headache
- fever.
Also read: Cicada Variant: Will The Current COVID Vaccine Provide Protection?
What Do The 75 Genetic Changes Mean?
Cicada, a descendant of the Omicron variant that emerged in 2021, was first detected in South Africa in 2024. However, it disappeared soon after, only to emerge early this year.
Prof Gupta told the publication that “the Cicada variant developed the 75 mutations to escape immunity, and it evolved by incubating a single patient for over a year”.
"This 'patient zero' would have been unable to clear the virus due to a compromised immune system," due to HIV or anti-cancer drugs.
Further, he explained that due to 75 mutations to the spike protein, the body’s antibodies will take a longer time to fight the Cicada variant.
However, there is no evidence yet that it is likely to cause more severe disease.
Will The Cicada Variant Cause Severe Illness?
The Cicada variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
Yet, the World Health Organization and health experts advise that existing COVID vaccinations can help prevent severe illness and hospitalization.
"It would appear that all the protections we have from our experience with the virus and with vaccines probably offer more limited—not zero—but more limited protection against this strain," Dr. William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center, was quoted as saying by Time.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
How To Safeguard
Dr. Sai Balasubramanian, a doctor and healthcare strategy executive, writing in Forbes, stressed the need to follow COVID practices such as masks and hand hygiene.
"Healthcare professionals recommend taking general precautions, similar to those used to prevent most viral transmission: get vaccinations when appropriate, wear masks in crowded areas or indoors where there is a high risk of transmission," he said.
He also urged “avoid individuals who have known illness or infections, wash hands frequently, and continue to stay apprised of local community guidelines and the infection spread”.
India To Strictly Inspect GLP-1 Drugs To Curb Misuse: Govt Flags Risks Amid Weight-loss Hype

Credit: iStock
GLP-1 drugs are powerful tools that can improve health, but it comes with risks and caveats. While these are medical therapies, they are being misused as cosmetic products by cosmetologists, physiotherapists, dermatologists, among others.
To curb its misuse and improve public health, the Indian government today issued guidelines on its use, while also flagging risks around it.
In an official statement issued today, the government has stated that it will roll out stricter inspections to curb the misuse of GLP-1 drugs in the country and will punish those who violate the rules.
“GLP-1 drugs are a significant medical breakthrough in treating type 2 diabetes and obesity, but they are not without risk. The drugs carry a wide range of side effects - from common symptoms like nausea and vomiting to severe complications including pancreatitis, kidney injury, and bowel obstruction. These risks make it essential that GLP-1 drugs are only taken under the supervision of registered medical practitioners/specialists,” the statement said.
In the wake of its hype over weight loss, the country's regulatory authorities have taken firm steps to prevent unsupervised use and malpractices in the supply chain.
It has “strongly advised” patients and the public to” consult a qualified medical specialist before use, and to obtain these medications with prescription only through legitimate, regulated channels”.
“Stricter inspections and surveillance will continue in the coming weeks. Businesses that break the rules will face license cancellation, fines, and legal action,” the statement said.
Also read: GLP-1 Drugs: Why India Needs Stricter Rules Now
What Are The Side-effects Of GLP-1 Drugs?
GLP-1 drugs work to keep food in the digestive system for longer – making people feel fuller for longer, thus reducing appetite and inducing weight loss. Hence, the drugs are also prescribed to people with obesity.
However, when done without clinical oversight, it can lead to severe health complications.
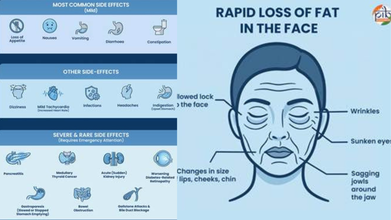
There are various side effects to taking GLP-1 drugs, including nausea and dizziness, to pancreatitis and medullary thyroid cancer.
Some serious side effects include
- Pancreatis
- Thyroid Cancer
- Acute (Sudden) Kidney Injury
- Worsening Diabetes-Related Retinopathy
- Gallstone & Bile Duct Blockage
- Rapid loss of fat in the face
- Wrinkles
- Sunken eyes
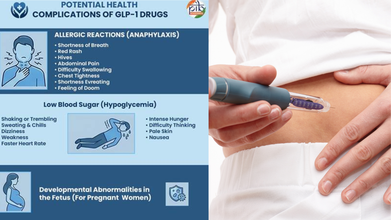
GLP-1 medications may also complicate various health conditions, such as allergic reactions, with shortness of breath and low blood sugar.
Rush For Generic Weight Loss Drugs
The government's advisory comes as patent for semaglutide -- an active ingredient in diabetes and anti-obesity drugs, specifically Wegovy and Ozempic -- expired on March 20.
This has led to a rush among Indian pharma companies to launch cheaper generic versions, significantly increasing affordability and access for millions battling Type 2 diabetes and obesity.
Also read: CDSCO threatens action against pharma companies for promoting GLP-1 weight-loss drugs
Major Indian drugmakers who launched their generic semglutide injection in the country in March include Sun Pharmaceutical Industries, Zydus Lifesciences, Alkem Laboratories, and Dr. Reddy’s Laboratories.
WHO Guidelines on GLP-1 drugs
Amid the increasing prevalence of GLP-1 drugs, the World Health Organisation (WHO), late last year, acknowledged its role in treating obesity.
However, it warned that medications like GLP-1 alone will not solve the problem affecting more than one billion people worldwide.
The global health body also issued conditional recommendations for using these therapies as part of a comprehensive approach that includes healthy diets, regular physical activity, and support from health professionals.
© 2024 Bennett, Coleman & Company Limited

