- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
This Woman Could Not Stay Sober Without Drinking- Until Doctors Found Her Bladder Was Brewing Its Own Alcohol

Image Credit: Health and me
It can be incredibly frustrating being constantly told that you have a drinking problem when you've never had a drop but for this 61-year-old Pennsylvania woman, each and every effort to become eligible for a life-saving liver transplant was thwarted by an insurmountable hurdle—she kept testing positive for alcohol. No matter how many times she asserted that she had not been drinking, her urine drug screens read otherwise. Accordingly, she was refused entry on the liver transplant waiting list numerous times, the doctors presuming she was struggling with alcohol addiction.
But the reality was much stranger than anyone might have dreamed. In what seems to be a first in medical history, physicians found that her own bladder was secreting alcohol—a process so unusual that it had not been given an official name yet. Her case sheds new light on how the human body, under abnormal circumstances, can simulate effects of alcohol use without ingestion.
The patient had severe liver cirrhosis, a scarring and liver function loss that requires a transplant to save her life. She also had diabetes that was uncontrolled, leading to high levels of glucose (sugar) in her urine.
Initially, physicians at one hospital brushed off her assertion that she was sober, believing that she was denying alcohol addiction. When she subsequently reported for assistance at the University of Pittsburgh Medical Center (UPMC) Presbyterian Hospital, the same problem persisted. Her urine samples consistently registered the presence of ethanol, the beverage type of alcohol, further increasing the medical staff's suspicion. Yet there was a critical discrepancy: whereas her urine tested positive for ethanol, it was free of the usual metabolic byproducts—ethyl glucuronide and ethyl sulfate—seen days after drinking.
This contradiction interested Dr. Kenichi Tamama, a senior researcher on the study and an associate professor of pathology and medical director of UPMC's Clinical Toxicology Laboratory. If the woman was consuming alcohol, the metabolites should have been found in her urine. Also, her blood alcohol tests were negative, and she never showed any symptoms of being intoxicated.
Following more research, the doctors discovered a surprising cause: yeast in the woman's bladder was fermenting sugar into alcohol. The process, normally related to making beer or wine, was occurring within her body. Consequently, they suggested calling her condition "urinary auto-brewery syndrome" or "bladder fermentation syndrome."
This condition is similar to, but different from, a rare disorder called auto-brewery syndrome (ABS), also referred to as gut fermentation syndrome. ABS happens when some microbes in the gut ferment carbohydrates into alcohol, leading to people with the condition becoming intoxicated without consuming alcohol. In the case of the Pennsylvania woman, however, her condition was confined to her bladder, so the alcohol did not reach her bloodstream. She therefore never seemed drunk, although her urine was ethanol-positive.
To be sure about their suspicion, scientists performed an experiment. The researchers took a fresh urine specimen from the patient and incubated it at a temperature of the human body (98.6°F or 37°C). Ethanol in the sample zoomed within no time. The sample did not yield any alcohol, however, if it were incubated below normal temperature (39°F or 4°C) or after the addition of a chemical for stopping fermentation. This test conclusively established that the yeast in her bladder was the cause of the alcohol production.
Additional lab tests determined the offender—Candida glabrata, a yeast that is normally present in the human microbiome. Candida glabrata is closely related to brewer's yeast, which is employed in alcoholic fermentation. Under conditions of excess sugar, as in the case of the woman with uncontrolled diabetes, Candida glabrata metabolized the glucose in her bladder to ethanol.
Due to these observations, the woman was finally reevaluated for liver transplant, a move that could turn around her condition and save her life. Her case highlights the need to cast aside standard assumptions of diagnosis and to explore infrequent metabolic phenomena.
Although sporadic reports of such cases have appeared, they were usually considered postmortem changes or laboratory artifacts due to the improper storage of urine samples. This case presents tangible evidence that bladder fermentation is possible in living patients and serves to increase awareness among medical professionals regarding distinguishing between true alcohol intake and endogenously produced alcohol.
What Is Auto-Brewery Syndrome?
Auto-brewery syndrome, or gut fermentation syndrome, is a rare condition where carbohydrates ferment to alcohol within the gastrointestinal tract. Patients can present with signs of intoxication, such as dizziness, slurred speech, and coordination impairment, without alcohol consumption. This results from an imbalance in gut microbiota, typically yeasts such as Candida.
The syndrome is well-documented in medical literature for more than 50 years, yet it remains significantly underdiagnosed. A study published in 2021 estimated that fewer than 100 cases had been reported globally. Experts, however, estimate that numerous other people might be suffering from the condition unknowingly, as the symptoms might be confused with alcohol intake, metabolic conditions, or psychiatric issues. In severe instances, people with ABS have been charged with DUI, even though they consumed no alcohol.
How Is Urinary Auto-Brewery Syndrome Different From Typical Auto-Brewery Syndrome?
Urinary auto-brewery syndrome (bladder fermentation syndrome) and conventional auto-brewery syndrome (gut fermentation syndrome) are similar but differ significantly.
In urinary auto-brewery syndrome, alcohol is synthesized in the bladder because of microbial fermentation of sugar. It may also happen in an open sample of urine outside the refrigerator. But as the alcohol is contained within the bladder and does not get into the bloodstream, individuals who have it do not have symptoms of being intoxicated.
In contrast, classical auto-brewery syndrome happens within the gastrointestinal tract, where fermentative microbes metabolize carbohydrates into alcohol. This alcohol is absorbed into the bloodstream, resulting in a positive plasma ethanol reading and creating symptoms of intoxication similar to that following alcohol intake.
Florida Restores H.I.V. Drug Access With 31 Million Emergency Funding

Credits: Canva and Wikimedia Commons
Florida on Tuesday approved nearly $31 million in short-term funding for H.I.V. medication for residents This is a reverse course after the state health officials restricted the access to a program that helps only those who could afford costly medications.
What Happened In The Past?
The Florida Department of Health had imposed tougher eligibility rules on March 1 that left 12,000 residents with H.I.V. without the access to medication covered by the state's AIDS Drug Assistance Program. This deprived them of life saving drugs, noted a nonprofit advocacy group - AIDS Healthcare Foundation.
The foundation also sued the department over these changes with protestors rallying in opposition at the State Capitol in Tallahassee. The department cited a projected $120 million state budget that restricted eligibility for the program.
Read: HIV vs. AIDS: What You Should Know About These Commonly Confused Terms
Yet, the Florida House of Representatives and State Senate, both led by Republicans, unanimously approved legislation that provides $30.9 million to fund the program through June 30 and restore the eligibility rules. Gov. Ron DeSantis, a Republican, signed the bill into law on Tuesday.
“Lawmakers on both sides understood the urgency of the crisis,” State Senator Carlos Guillermo Smith, a Democrat from Orlando who spoke out in support of the bill on the floor, said in an interview on Wednesday. “It is a matter of life and death. People had already lost access to their medications.”
Restriction On AIDS Drug Assistance Programs
As per an analysis by KFF - a health research group, Florida was one of the nearly 20 states, led both by Republicans and Democrats that imposed restrictions on AIDS Drug Assistance Programs.
The programs cover the cost of H.I.V. medications, which can run into thousands of dollars each month. They provide drugs free to some individuals and help others by paying their insurance premiums. Overall, they support about a quarter of the 1.2 million people living with H.I.V. in the United States.
However, these programs are under growing pressure. The cost of H.I.V. medications continues to rise, and the expiration of health care subsidies has led to a sharp increase in insurance premiums. Despite this, federal funding for these programs has remained largely unchanged for more than a decade.
In Florida, the Department of Health had reduced eligibility for the state’s AIDS Drug Assistance Program to 130 percent of the federal poverty level, which is about $20,748 a year for an individual, down from 400 percent or $63,840. A new law has now reversed this change, restoring eligibility back to the 400 percent threshold.
“For 10 weeks, 12,000 Floridians living with H.I.V. did not know if they could fill their next prescription,” Esteban Wood, the director of advocacy and legislative affairs at the AIDS Healthcare Foundation, said in a statement on Tuesday. “Today, they can.”
US FDA Approves Drug To Treat Rare Childhood Syndrome
Credit: Wikimedia Commons
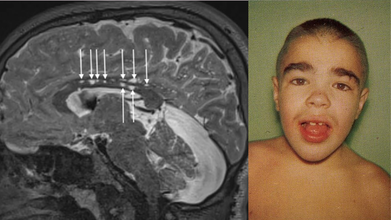
The US Food and Drug Administration (FDA) has approved a drug therapy to address neurologic symptoms of a rare genetic disorder — Hunter syndrome.
The X-chromosome-linked disease, occurring predominantly in males, has an estimated 2,000 affected individuals worldwide.
The drug Avlayah, developed by pharma company Denali Therapeutics, targets certain individuals with Hunter syndrome (Mucopolysaccharidosis type II or MPS II).
It is the first therapy to address the neurologic complications of Hunter Syndrome, such as
- progressive cognitive decline,
- behavioral problems,
- hydrocephalus,
- spinal cord compression,
- seizures,
- carpal tunnel syndrome.
“Today is a milestone day for children and their families battling Hunter syndrome,” said FDA Commissioner Marty Makary, in a statement.
“Avlayah is the first product approved to address neurologic complications of Hunter Syndrome, a very rare and often severe X-linked disorder in children, affecting about 500 people in the US, almost exclusively males,” added Acting CDER Director Dr. Tracy Beth Hoeg.
The FDA noted that Avlayah, the once-weekly drug given via IV infusion, must begin in presymptomatic or symptomatic pediatric patients weighing at least 5 kg before advanced neurologic impairment.
What Is Hunter Syndrome?
Hunter syndrome is a rare inherited lysosomal disorder in which sugar molecules called glycosaminoglycans build up within the cells’ lysosomes.
The condition affects physical and mental development and causes abnormalities in the skeleton, heart, respiratory system, brain, and other organs.
Hunter syndrome is a rare congenital metabolic disease. It was first reported in 1917 by a Canadian physician, Charles Hunter, in two brothers in a family.
The brothers presented typical signs, such as
- short stature,
- inguinal hernia,
- macroglossia,
- enlarged skull,
- decreased hearing,
- coarse facial features,
- protruded abdomen with hepatosplenomegaly,
- umbilical hernia,
- skeletal deformities.
The younger brother had symptoms of Central Nervous System (CNS), including seizures and cognitive decline, while the older brother did not have CNS involvement.
The estimated incidence is 1 in 162,000 live male births.
How The FDA Approved Avlayah
The FDA approval came after Avlayah showed promise in reducing cerebrospinal fluid heparan sulfate — one of the glycosaminoglycans that accumulates in the body in this disorder and is linked to the organ damage that occurs in early childhood.
The phase 1/2 multi-cohort, single-arm, open-label trial enrolled 47 pediatric patients with Hunter syndrome aged 3 months to 13 years.
Of these, 44 patients with measurements at had a 91 percent average decrease from baseline in CSF.
Denali is now conducting a randomized clinical trial that is more than 95 percent enrolled to evaluate the clinical benefit of this product.
"In the meantime, families with young children with Hunter Syndrome will have access to a product that may favorably alter the course of the disease at the crucial time in life when there is the greatest potential for benefit," Hoeg said.
Are There Any Side Effects?
Avlayah’s labeling includes a boxed warning for allergic reactions, including anaphylaxis, associated with the drug.
The FDA recommended that patients start therapy in a health care setting with appropriate medical monitoring and support measures.
The common side effects of Avlayah include
- upper respiratory tract infection,
- ear infection,
- fever,
- anemia,
- cough,
- vomiting,
- diarrhea,
- rash,
- COVID-19,
- runny nose,
- nasal congestion,
- fall,
- headache,
- skin abrasion,
- hives.
The FDA also suggested that healthcare workers monitor
- hemoglobin levels due to the risk of anemia,
- kidney function and urine protein levels due to
- the risk of membranous nephropathy (a kidney disease).
CDSCO Launches National Crackdown On Unapproved Medicines

Credit: Canva, CDSCO
The Central Drugs Standard Control Organisation (CDSCO) has now launched a nationwide crackdown on unapproved Fixed Dose Combination (FDCs) after 90 combination medicines that were being sold without mandatory central approval were busted last week.
CDSCO, India's apex drug regulatory body in the country, made the shocking discovery after scrutinizing drug samples uploaded on the SUGAM portal, the government’s online system for drug testing data.
Several of these samples were found to fall under the ‘new drug’ category but lacked approval from the central authority. As a result, the CDSCO wrote a letter to all states and union territory drug controllers, asking them to investigate manufacturers and marketers of these FDCs.
The letter reads: "The SUGAM lab testing data for the year 2025, a large number of drug samples (FDCs) are detected as unapproved and fall under the category of 'New Drug'. No new drug shall be manufactured for sale unless it is approved by the Licensing Authority 'as defined in Rule 3 of New Drugs and Clinical Trial Rules, 2019, Further, as per Rule 80 of New Drugs & Clinical Trial Rules 2019, a person who intends to manufacture new drug in the form of API or Pharmaceuticals formulation, as the case may be, for sale or distribution, shall make an application for grant of permission to the Central Licensing Authority in Form CT-21 along with a fee as specified in Sixth Schedule."
"The presence of unapproved drugs in the supply chain is a matter of serious concern, posing potential risks to public health and safety. It also indicates non- compliance with the provisions of the Drugs and Cosmetics Act, 1940, and rules made thereunder.
"Accordingly, please initiate appropriate investigation and regulatory action against the concerned manufacturers, marketers, and other stakeholders, as deemed fit. Kindly ensure strict monitoring and enforcement to prevent manufacture, sale, and distribution of such unapproved drugs," the letter states, on appropriate action against the sale and distribution of unapproved drugs.
FDCs are drugs that contain two or more active ingredients in a single formulation. The CDSCO has asked states to investigate and submit reports on the action taken at the earliest.
What Made It To The Unapproved List Of FDCs?
Cough And Cold Medicines
Cough and cold combination medicines have formed the single largest group. At least 14 to 16 entries show combination of ingredients found in everyday pharmacy shelves: dextromethorphan, ambroxol, guaiphenesin, chlorpheniramine, phenylephrine, terbutaline, and menthol in various permutations.
Several of these are sold as flavoured syrups for children. Budesonide-levosalbutamol inhalation suspension that is used for asthma and chronic obstructive pulmonary disease, too features in the list.
Nutritional Supplements
The next group is of the vitamins, minerals and other nutritional supplement which has roughly 18 to 20 entries. They include:
- Multivitamin capsules
- Iron-folic acid syrups
- Calcium-vitamin D3-K2 combinations
- Abapentin-methylcobalamin preparation
This group has accounted for at least 10 entries, which include antifungal creams that could lead to potential misuse and skin damage when sold without prescriptions.
Anti-Diabetic Medicines
This has accounted for six entries, and the combination includes:
- Glimepiride paired with metformin or pioglitazone
What Is India's Rule For FDCs?
Under India's New Drugs and Clinical Trial Rules of 2019, any FDC is treated as a new drug and must have the central government's approval before it could be manufactured and sold. The regulator's letter noted that there were presence of unapproved drugs in the supply chain that could pose risks to public health. This also amounts to a violation of the Drugs and Cosmetics Act, 1940.
“The presence of unapproved drugs in the supply chain is a matter of serious concern, posing potential risks to public health and safety. It also indicates non- compliance with the provisions of the Drugs and Cosmetics Act, 1940, and Rules made thereunder," it said.
DCGI urged states and UTs "to examine the unapproved FDCs in generic name as mentioned in the attached annexure". This is done to check whether any approval has been granted by the local office. "Accordingly, please initiate appropriate investigation and regulatory action against the concerned manufacturers, marketers, and other stakeholders, as deemed fit. Kindly ensure strict monitoring and enforcement to prevent the manufacture, sale, and distribution of such unapproved drugs," the letter noted.
© 2024 Bennett, Coleman & Company Limited

