- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
US FDA Approves First At-Home Device To Treat Depression – Here’s What You Need To Know

The U.S. Food and Drug Administration has cleared an at-home brain stimulation headset developed by Flow Neuroscience to help treat depression, giving patients another option beyond standard antidepressant drugs, which many people struggle to stay on because of long-term side effects, the company announced on Thursday.
Rates of depression in the U.S. have climbed sharply, rising by about 60% over the past ten years and now affecting more than 20 million adults, according to data from the Centers for Disease Control and Prevention.
FDA Approves First At-Home Brain Stimulation Device For Treatment of Depression
The US Food and Drug Administration (FDA) has approved a home-use brain stimulation device designed to treat major depressive disorder in adults. The device, known as FL-100 and made by Flow Neuroscience, is the first at-home treatment of its kind to receive approval in the United States.
The FDA’s decision was supported by results from the Empower phase 2 clinical trial, which found that 58% of participants reached remission after 10 weeks of treatment. Flow Neuroscience also reported that among users worldwide, 77% noticed an improvement in symptoms within three weeks. To date, more than 55,000 patients have used the device in regions including Europe, the UK, Switzerland, and Hong Kong.
Brain Stimulation Device For Depression: How Will The Headset Work?
The prescription device, called FL-100, uses transcranial direct current stimulation to send a mild electrical current to the brain’s prefrontal cortex, an area involved in mood control and stress response. According to Flow, this part of the brain is often less active in people living with depression, a condition that affects over 20 million adults across the U.S.
Flow noted that roughly one in three people with depression does not respond adequately to antidepressant medications, and many others discontinue them because of unwanted side effects.
“Flow’s FDA approval marks a turning point in depression care, representing the first real move away from medication-only treatment toward technology-based therapies with fewer side effects,” said Flow CEO Erin Lee in a statement.
During the study, participants wore the headset for 30 minutes per session while resting, beginning with five sessions a week for the first three weeks, then reducing to three sessions per week for the rest of the trial. The program also included live remote monitoring through video calls.
Researchers observed benefits in patients who were already taking antidepressants for treatment-resistant depression or receiving psychotherapy, as well as in those who were not using medication at all.
“Among people using Flow in real-world settings, 77% report feeling better in as little as three weeks,” said Kultar Garcha, M.D., chief medical officer at Flow. “A treatment that was once limited to clinics can now be used at home, offering a practical way to widen access to effective depression care.”
Flow said it plans to launch the headset commercially in the U.S. in the second quarter of next year and intends to explore its potential use for conditions such as traumatic brain injury, addiction, and sleep disorders.
“We want Flow to become part of the standard pathway for treating depression,” Lee said. “Our focus is clear: to help as many people as possible reach remission and regain their quality of life.”
Leucovorin Approved By FDA But For A Rare Genetic Disease, Not For Autism
Credits: Reuters
The US Food and Drug Administration approved leucovorin, but not for what White House had recommended for in its September briefing. FDA has approved the prescription drug leucovorin for cerebral folate deficiency in the receptor 1 gene, a genetic condition that is estimated to affect only about 1 in 1 million people.
In September, the White House touted leucovorin as a potential treatment for children with autism. Health and Me previously reported on how parents were struggling to find the prescription drug, while not enough scientific evidence was there to support the claim.
What Is Leucovorin?
It is a high-dose B vitamin, which is commonly used as a treatment to counteract the side effects of chemotherapy. It has been approved by the FDA for cerebral folate deficiency in the receptor 1 gene. The disease is rare and fewer than 50 cases have been identified worldwide. FDA Commissioner Dr Marty Makary called this approval "a significant milestone" for patients with the condition.
However, in September last year, Markary and US Health and Human Services Secretary Robert F Kennedy Jr said that the drug could help children with autism. Kennedy, standing next to US President Donald Trump, said, they had "identified an exciting therapy that may benefit large number of children who suffer from autism."
CNN reported that in Austin, Texas, Meagan Johnson, a mother with an autistic son of three years of age, spent four days calling pharmacies across the region searching for leucovorin. She contacted nearly 40 pharmacies around her home in Pflugerville, hoping to locate the medication. Health and Me reported how getting the prescription turned out to be far harder than obtaining it.
This update on leucovorin, notes CNN have hit some families hard, especially the families of children with autism. This is because of the September briefing and when Trump also said that painkiller Tylenol could in fact cause autism. The drug leucovorin was used as something that could give them hope, however, with the update in its label, parents efforts to find the prescription drugs could feel like in vain.
The Rare Condition For Which Drug Got Its Approval
The drug got its approval for folate deficiency in the receptor 1 gene. This is a defect that causes proteins that guide folate into the brain to malfunction. As a result, normal levels of folate is found in the blood, but not in the brain or nervous system, due to which tasks like thinking, speech and movement could not be performed.
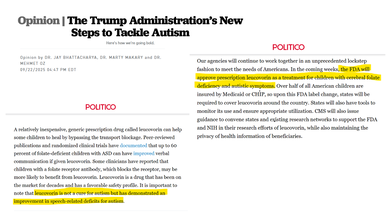
In an op/ed published in Politico and reposted on the White House website last fall, Markary said that his agency would "approve prescription leucovorin as a treatment for children with cerebral folate deficiency and autistic symptoms". However, he did not that it cause not a cure for autism, but there have been studies that show that it could help children with folate deficiency and autism to improve speech by roughly 60 per cent. The Trump administration made a bold move to expand the usage of the drug by "opening the door to the first FDA-recognized treatment for autism." wrote Markary and other HHS leaders.
AIIMS And ISRO Sign MoU To Advance Research In Space Medicine

Credits: Dr P Sarat Chandra (LinkedIn)
All India Institute of Medical Sciences (AIIMS) and the Indian Space Research Organisation (ISRO) signed a Memorandum of Understanding (MoU) on Monday to collaborate on advancing research in space medicine.
The MoU was signed between M Srinivas, Director (AIIMS), New Delhi and Dinesh Kumar Singh, Director, Human Space Flight Centre (HSFC), ISRO. The event brought together senior leadership from AIIMS and ISRO, including ISRO Chairman V Narayanan and Secretary, Department of Space, as well as Deans, Heads of Departments, faculty members, and studies from AIIMS New Delhi.
Also Read: Is There A Link Between Your Kidney Health And Other Chronic Diseases? Study Says Yes
As per the news agency Asian News International (ANI), a press release of the collaboration noted for a cooperative framework for ground-based and space-based research in Space Medicine. The press release also noted that the research will cover areas like human physiology, cardiovascular and autonomic regulation, musculoskeletal health in microgravity, microbiome and immunology, genomics and biomarkers and behavioral health.
“This MoU will give us the escape velocity to venture together into the field of space medicine. Collaborative research between AIIMS and ISRO will benefit patients, the nation, and ultimately humankind. As India moves toward Viksit Bharat by 2047, we hope to emerge as a Vishwaguru in space medicine as well,” said Prof M Srinivas, Director, AIIMS, New Delhi.

Chairman of ISRO Dr V Narayanan and Secretary, Department of Space also highlighted India’s remarkable journey in space programme. He noted how India evolved from the time when rockets and equipment were transported on bicycles and bullock carts to today, where it is going to be leading medical and research institutions like AIIMS to strengthen India’s human spaceflight capabilities.
The programme also featured a presentation on AIIMS New Delhi’s ongoing work in space medicine research, delivered by Prof. KK Deepak, former Head of the Department of Physiology. The event was attended by Heads of Departments and faculty members from across AIIMS New Delhi, along with representatives from the Resident Doctors Association (RDA), the AIIMS Student Association (ASA), and the Society of Young Scientists (SYS).
The collaboration represents an important step toward enhancing India’s capabilities in human spaceflight and biomedical research. It also aligns with the country’s broader scientific ambitions as India moves toward its centenary in 2047 and the vision of Viksit Bharat.
What Do We Already Know About Space Medicine?
As per a 2017 study in the British Journal of Anaesthesia, space medicine is fundamental to the human exploration of space. It supports survival, function and performance in this challenging as potentially lethal environment. It is international, intercultural and interdisciplinary, operating at the boundaries of exploration, science, technology and medicine.
Space medicine specialists play a crucial role in safeguarding astronauts and other spaceflight participants. Known as “flight surgeons,” they help develop strategies to protect the health, safety, and performance of space travellers in the extreme conditions of space. Their responsibilities span every stage of a mission — from selection and training to in-flight care, post-mission rehabilitation, and monitoring long-term health effects.
With inputs from ANI
Virus Without Vaccine Hits California; No Need To Worry, Say Public Health Officials
Credits: Canva
Sick days are again piling up with a respiratory disease, not COVID-19 or the flu. This disease is most likely the one Americans have not heard of. This is HMPV or human metapneumovirus. Doctors have asked people to stay vigilant as seasonal flu virus could lead to pneumonia and bronchitis, and it is spreading in California wastewater and around the country. However, as per public health officials, there is nothing to get worried at this point.
Also Read: Is There A Link Between Your Kidney Health And Other Chronic Diseases? Study Says Yes
As per the public database WasterwaterScan Dashboard, high levels of HMPV were detected across Northern California cities. The highest levels were reported in Redwood City, whereas elevated levels were found in San Francisco Bay Area and Napa's Wine Country. What's more dangerous is that this virus is without a vaccine.
The good news is that in other parts of country HMPV remains lower. However, the Centers for Disease Control and Prevention (CDC) noted that data from October 2025 shows the cases are trending up, especially during winter and spring.
Dr. Matthew Binnicker, director of the Clinical Virology Laboratory at Mayo Clinic, as reported by The Independent said, "In the late winter, early spring, it can account for five percent to 10 percent of all the respiratory infections that we diagnose in the United States. So it's definitely out there." Experts explain that other viruses like HMPV or influenza get a chance when COVID is quieter.
Read: HMPV Virus Cases Surging In California, New Jersey: Is It Dangerous?
Virus Without A Vaccine: What Is HMPV?
HMPV was first discovered in 2001 and is part of the Pneumoviridae family along with the Respiratory syncytial virus (RSV).
HMPV most likely spreads from an infected person to others through:
- the air by coughing and sneezing
- close personal contact, such as touching or shaking hands
- touching objects or surfaces that have the viruses on them, then touching the mouth, nose, or eyes
In the US, HMPV circulates in predictable patterns each year, typically beginning in winter and lasting through spring.
The usually self-limiting and mild symptoms typically last 4 to 5 days. These include:
- cough
- fever
- nasal congestion
- shortness of breath
People at risk include:
- elderly people
- children
- people with comorbidities
"The HMPV is not deadly, and there is no evidence of mortality or a severe transmission rate," former Indian Council of Medical Research (ICMR) scientist, Dr. Raman Gangakhedkar, told IANS, during the virus's outbreak in India in 2025.
“The virus may cause pneumonitis-like illness, but the mortality rates are almost unknown so far. HMPV has a global prevalence of about 4 per cent,” he added.
While no vaccine or treatment can prevent HMPV infection, to avoid the infection, individuals must:
Practice good hygiene and cover your mouth and nose with a tissue when coughing or sneezing, or use your elbow, not your hands, for it. And wash your hands properly, especially in healthcare settings.
Why Are People Hearing About The Virus Without Vaccine Now?
The reason people have not heard about it before is because its symptoms are nearly indistinguishable from other respiratory infections, so often it could go undiagnosed. Furthermore, earlier the COVID cases and influenza cases sparked up, which deviated the attention from HMPV. Now, with the season gone, and increased awareness around respiratory illnesses, greater attention to viruses like HMPV is also paid.
© 2024 Bennett, Coleman & Company Limited

