US FDA Approves Drug To Treat Rare Childhood Syndrome
Credit: Wikimedia Commons
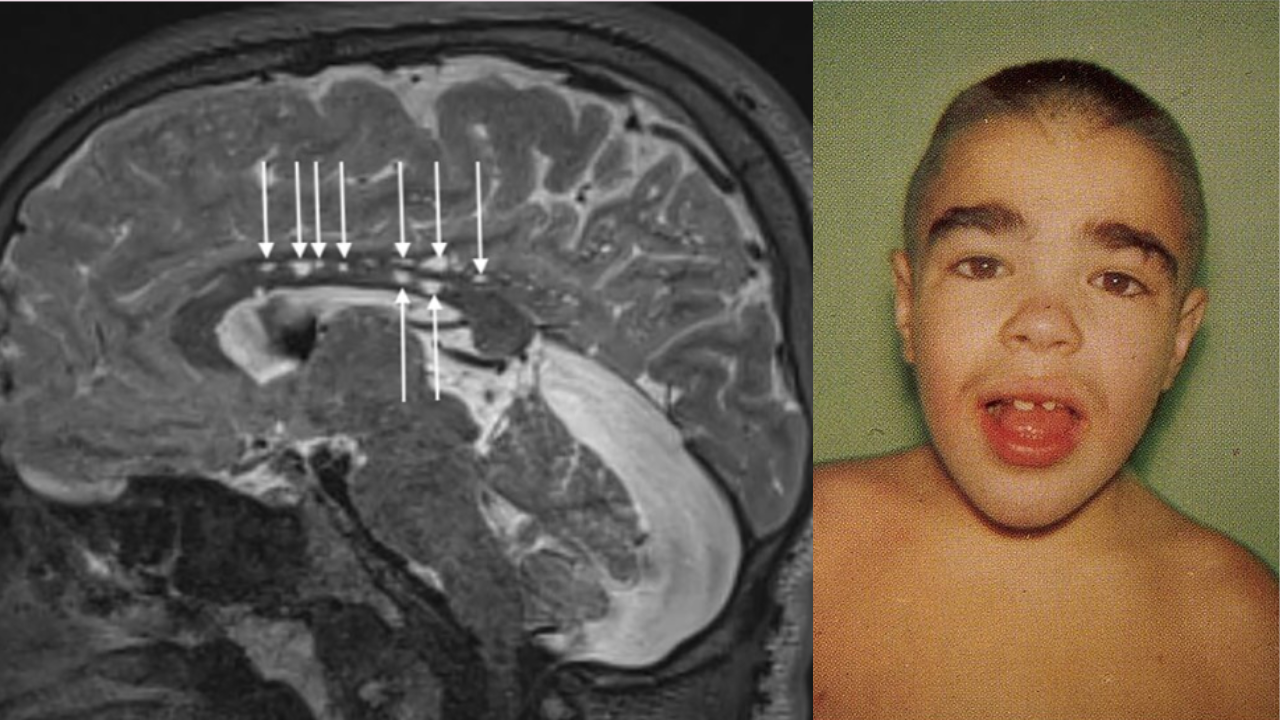
SummaryThe drug Avlayah, developed by pharma company Denali Therapeutics, addresses the neurologic complications of Hunter Syndrome, which occurs predominantly in males. It has an estimated 2,000 affected individuals worldwide.
End of Article
