- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
US FDA Recalls Broccoli Amid The Risk Of Listeria Outbreak

Credits: Canva
The US Food and Drug Administration (FDA) issued a warning for broccoli sold at Walmart and recently has elevated the risk for the same, This has been the highest recall of FDA, said the reports. The FDA said, "This advisory is being initiated due to possibility of contamination with Listeria monocytogenes. The potential for contamination was discovered during random sampling by Texas Health & Human Services from a Texas store location where one of multiple samples yielded a positive test result."
What is Listeria Monocytogenes?
This is a bacterial pathogen which is responsible for listeriosis. This is a serious foodborne illness. It thrives in various environment, including soil, water, and food processing facilities and is gram-positive and rod-shaped in its nature. It can also grow at refrigeration temperature which can then infect dairy products, deli meats, seafood, and unpasteurized milk.
There was one sick individual in each of New York and New Jersey, two in Illinois, and seven in California, including the dead child. According to the CDC, they all identify as Asians.
What Does Listeria Outbreak mean?
As per the CDC, Listeria are bacteria or germs that can contaminate many food and those who eat can get infected with the bacteria. CDC mentions that it is rare, however, could be serious, though there exists steps to prevent this infection.
It is a foodborne illness caused by the bacteria L. monocytogenes. Symptoms include fever, chills, and headache. It can cause invasive illness and intestinal illness. It is also the third leading cause of deaths from foodborne illness in the US, with 1,600 people infected each year, out of which 260 die.
Symptoms often appear within two weeks of consuming Listeria-contaminated food, but can appear as early as the same day or as late as ten weeks later.
A fever, muscle aches, and fatigue are common symptoms of pregnancy.
People who are not pregnant usually have fever, muscle aches, and tiredness. They may also get a headache, stiff neck, confusion, loss of balance, or seizures.
Is It Deadly?
It is deadly because the pathogen forms a layer of biofilm on the food which makes it resistant to cleaning or disinfectants. It also persists in food production environments, and increases the risk of contamination.
What can be done to be safe?
The best way to stay safe is by avoiding raw sprouts, soft cheeses made from unpasteurized milk, refrigerated deli meats or broccoli forets, unless heated to really hot temperatures.
Are There People more at risk?
Pregnant women, persons over the age of 65, and people with compromised immune systems are most vulnerable to Listeria. This is due to the fact that Listeria is more prone to move from the gut to other areas of the body, culminating in invasive listeriosis, a serious illness.
Listeria can cause pregnancy loss, early birth, or a life-threatening infection in newborns.
Listeria frequently causes hospitalization and, in some cases, death in those aged 65 and older or with a weakened immune system.
1st-Ever In-Womb Stem Cell Therapy for Spina Bifida Found Safe, Gets US FDA Backing: The Lancet
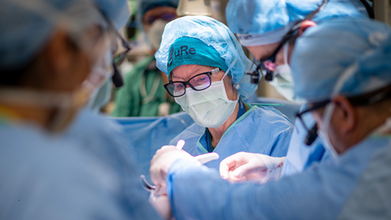
Credit: University of California-Davis Health
In a medical milestone, a team of US surgeons has deemed an in-utero surgery that adds stem cells to treat spina bifida -- a birth defect -- to be safe. This new type of fetal therapy does not just repair the defect but may also help heal and protect the baby’s developing spinal cord.
The Phase 1 clinical trial, published today in The Lancet and based on six babies, showed that adding a layer of human placenta-derived stem cells to standard fetal surgery can be done safely. The early safety results have prompted the US Food and Drug Administration (FDA), along with an independent monitoring board, to approve the next phase of the study.
Spina bifida, also known as myelomeningocele, occurs when spinal tissue fails to fuse properly during the early stages of pregnancy. The birth defect can lead to a range of lifelong cognitive, mobility, urinary, and bowel disabilities.
While surgeons have previously performed prenatal surgeries, this is the world’s first in-utero stem cell therapy for spina bifida. It is also the only clinical trial aimed at improving outcomes beyond those achieved with fetal surgery alone.
“Putting stem cells into a growing fetus was a total unknown. We are excited to report strong safety results,” said Diana Farmer, the clinical trial’s principal investigator and chair of the Department of Surgery at the University of California-Davis Health.
“It paves the way for new treatment options for children with birth defects. The future is exciting for cell and gene therapy before birth,” she added.
How The Breakthrough Fetal Surgery Was Conducted
Surgeons made a small opening in the mother’s uterus and gently positioned the fetus to expose its back and the spina bifida defect.
A small patch containing living stem cells was then placed directly over the exposed spinal cord before closing the layers of the back to allow the tissue to regenerate.
The stem cells, taken from donated placentas, are designed to protect the developing spinal cord from further damage before birth.
Treating Spina Bifida Before Birth
The findings from the first six babies in the trial, who were closely monitored from surgery through birth, revealed no safety concerns related to the stem cells.
After surgery, the babies experienced no infections or spinal fluid leaks. No abnormal tissue growth or tumors formed at the repair site.
All six surgeries were successful, and the stem cell patch was placed as planned in every case. All surgical wounds healed completely.
MRI scans also showed reversal of hindbrain herniation -- a condition commonly associated with spina bifida -- in all infants, an indicator of surgical success.
Hindbrain herniation in babies, often referred to as Arnold-Chiari II malformation, involves the lower part of the brain descending into the cervical spinal canal.
This can block the flow of cerebrospinal fluid, causing dangerous hydrocephalus (fluid buildup). None of the babies required a shunt for hydrocephalus before hospital discharge, the researchers said.
What Is Spina Bifida
Spina bifida affects about 1 in 2,500 births worldwide, with an estimated 1,500 to 2,000 babies diagnosed each year in the United States alone.
It is a common neural tube defect, with higher prevalence observed in regions with lower folic acid fortification. Open spina bifida specifically occurs in approximately 1 in every 2,875 births.
This surgical intervention aims to significantly improve motor function, and increase the likelihood of walking independently.
The first phase of the trial was funded by a $9 million state grant from the California Institute for Regenerative Medicine (CIRM).
The trial is now enrolling up to 35 patients in its Phase 1/2a study.
Children will be followed through age six to evaluate long-term safety and early signs of improved movement, as well as bladder and bowel function.
Indian Drugmakers Are Gearing Up To Launch Generic Semaglutide Injections As Weight-loss Drugs Patent Expires In March

Credits: Canva
Indian drugmaker Dr Reddy's Laboratories is now gearing up to launch its generic semglutide injection in the country in March under the brand name Obeda, reported the news wire agency Reuters. Patent protection for semglutide expires in India in March 2025. This has triggered a rush among Indian drugmakers to come up with lower-cost versions.
Semaglutide is the active ingredient in Danish drugmaker Novo Nordisk's Ozempic and Wegovy, popularly known as the weight loss drugs, but also effective for diabetes and used primarily for that.
Also Read: Scientists Develop First Antibodies To Block Epstein Barr Virus
Indian Drugmakers Rush For Generic Weight Loss Drugs
Hyderabad-based Dr Reddy's already applied for trademark with the brand name Obeda and a logo, reported Reuters, based on a government filing. In an email response to Reuters, Dr Reddy's spokesperson said, "As semaglutide is yet to be officially launched, it would not be appropriate to refer to or publish any name as the brand name at this stage."
Other companies like Sun Pharma, Zydus Lifesciences, and Nacto Pharma too are entering the rat race of launching multiple generic versions to make the treatment more affordable for patients with obesity and weight-related health risks.
Sun Pharma also announced the plans for "day-one" launches of generic prefilled pens.
Will Launching Generic Weight Loss Drugs Make It Cheaper To Buy?
Until the patent is expired, the semaglutide therapies are owned by the original company. In India, semaglutide injections like Ozempic and Wegovy are soled at a high cost that has limited accessibility to many patients.
Industry analysts, as reported by NDTV, expect that one generic semaglutide enters the market from March 21, 2026, prices could be cut roughly by 50 per cent as compared to the prices it started with. This means Wegovy which was previously sold for around Rs. 10,000 per month could fall somewhere between Rs 3,500 to Rs. 4,000 per month for starter doses.
Dr Reddy's Laboratories have positioned their generic brands competitively, and could potentially offer discounts of up to 50 to 60 per cent in early competition.
How Does Semaglutide Work?
Semaglutide works as a GLP-1 receptor agonist that mimics the GLP-1 hormone to regulate appetite and blood sugar. It slows gastric emptying and makes you feel fuller longer. It also signals the brain to reduce hunger and cravings, and triggers the pancreas to release insulin when blood sugar is high.
Read: Wegovy And Ozempic Will Cost Less In 2027, Novo Nordisk Slashes Weight Loss Drugs Prices By Half
They work by increasing insulin release in a glucose-dependent manner, decreasing the liver's production of glucagon, and slowing down the emptying of the stomach, which helps lower blood sugar levels after a meal. They also act on the brain to suppress appetite and increase feelings of fullness, leading to reduced calorie intake.
In people with type 2 diabetes, notes Harvard Health, the body's cells are resistant to the effects of insulin and body does not produce enough insulin, or both. This is when GLP-1 agonists stimulate pancreas to release insulin and suppress the release of another hormone called glucagon.
These drugs also act in the brain to reduce hunger and act on the stomach to delay emptying, so you feel full for a longer time. These effects can lead to weight loss, which can be an important part of managing diabetes.
Scientists Develop First Antibodies To Block Epstein Barr Virus
Credits: Canva
Scientists for the first time ever developed antibodies that could block Epstein Barr virus or EBV. This is a pathogen that is estimated to infect 95 per cent of the global population. The Epstein Barr virus is also linked with multiple types of cancer, neurodegenerative diseases and other chronic health conditions.
Antibodies for Epstein Barr Virus: How Does It Work?
Scientists used mice with human antibody genes. The team then developed new genetically human monoclonal antibodies that would prevent two key antigens on the surface of the Epstein Barr virus from binding to entering human immune cells.
The findings are published in Cell Reports Medicine. The study shows how the newly identified monoclonal antibodies successfully block infection in mice with human immune systems when they had EBV.
Scientists View On The Antibodies That Block Epstein Barr Virus
"Finding human antibodies that block Epstein Barr virus from infecting our immune cells has been particularly challenging because, unlike other viruses, EBV finds a way to bind to nearly every one of our B cells," explained Andrew McGuire, Ph.D., a biochemist and cellular biologist in the Vaccine and Infectious Disease Division at Fred Hutch. "We decided to use new technologies to try to fill this knowledge gap and we ended up taking a critical step toward blocking one of the world's most common viruses."
What Was the Challenge In The Creation Of Antibodies To Block Epstein Barr Virus?
The key challenge was to pursue human monoclonal antibodies that could actually halt the Epstein Barr virus infection without triggering an anti-drug response to the antibodies themselves. This is a common response among patients who are treated with antibodies raised in other animals.
The researchers targeted two antigens namely: gp350, which helps EBV bind to cell receptors. The second antigen was gp42 that allows EBV to enter and infect human antibody genes. This has led to the monoclonal antibodies to work against gp350 and eight gp42.
"Not only did we identify important antibodies against Epstein Barr virus, but we also validated an innovative a new approach for discovering protective antibodies against other pathogens," noted Crystal Chhan, a pathobiology Ph.D. student in the McGuire Lab. "As an early-career scientist, it was an exciting finding and has helped me appreciate how science often leads to unexpected discoveries."
Read: Chronic Epstein-Bar Virus: Can A Common Viral Infection Cause Fatal Brain Damage?
What Is Epstein Bar Virus (EBV)?
EBV is a highly contagious virus that can pass through bodily fluid contact. It is the cause of one of the most common infections, mononucleosis or mono. Once you get this virus, it stays in your body, inactive and in a dormant state until it becomes reactivated.
According to the Frontiers in Immunology, Chronic active Epstein-Barr virus (CAEBV) disease is a very uncommon problem where people's bodies can't fight off the EBV virus. It keeps getting worse, with a lot of the virus's DNA in their blood and the virus attacking their organs with infected blood cells.
People with this disease often have fevers, swollen glands, a big spleen, liver problems caused by EBV, or low blood counts. Over time, their body's ability to fight off infections gets weaker. If they don't get treatment, they can die from other infections, a problem where the body attacks its own blood cells, organ failure, or cancers linked to EBV.
The only treatment that has been proven to work for this disease is a stem cell transplant (a procedure to replace damaged blood cells with healthy ones). Right now, scientists are trying to figure out why this disease happens. They are looking at problems with the body's defense system (immune defects) and changes in people's genes that might be linked to the disease. A new study as also revealed that EBV may also cause us brain damage due to a disease called encephalitis.
Symptoms Of Epstein-Bar Virus
EBV infections do not cause symptoms especially for children, teens and adults are more likely to experience symptoms like fever, feeling tired or fatigued headache, sore throat, swollen lymph nodes in your neck and arm, enlarged spleen or swollen liver, body aches and skin rash. The symptoms of this usually last for two to four weeks. Things are a bit different with Chronic EBV. In rare cases of EBV, it can lead to a chronic condition called active EBV (CAEVB) some symptoms include, swollen and tender lymph nodes, fever, enlarged liver, fatigue, sore throat, headache, muscle pain joint stiffness, anemia and liver failure.
© 2024 Bennett, Coleman & Company Limited

