- Health Conditions A-Z
- Health & Wellness
- Nutrition
- Fitness
- Health News
- Ayurveda
- Videos
- Medicine A-Z
- Parenting
FDA Approves New COVID-19 Vaccines In US With Restricted Access, Check Eligibility And Other Rules
Credits: iStock
The US Food and Drug Administration (FDA) has licensed updated COVID-19 vaccines but imposed new limits that limit who can get them. The move is a turning point in pandemic policy, limiting vaccinations mainly to those most at risk of severe illness—older adults and those with underlying health conditions. For the first time since the pandemic began, the U.S. government is no longer advising mass COVID-19 vaccinations.
"Today's framework provides science, safety, and common sense," declared Health and Human Services Secretary Robert F. Kennedy Jr. on social media, adding that vaccines are still accessible to everybody who decides to take them after checking with a doctor.
Also Read: Can You Test Negative For Covid And Still Be Infected?
But Kennedy's past vaccine skepticism and his administration's actions to reduce funding for vaccine research have come under fire. He has already questioned the safety of vaccines for COVID and flu, and his agency's rulings on occasion have overridden the advice of career scientists.
During most of the pandemic, COVID shots had been available for all people six months and older. This universal availability was intended to slow spread and avoid severe illness. Healthy young adults and healthy children without medical issues might find it difficult to get vaccinated under the new limitations, particularly in anticipation of the predicted winter surge.
The Centers for Disease Control and Prevention (CDC) will eventually come forward with its recommendations, but preliminary indications are a relaxation of routine vaccination in healthy children and pregnant women. Public health professionals have sounded an alarm, suggesting that policy changes place some segments of the population at risk.
Who Can Get the New Vaccines?
In the new FDA framework, COVID vaccines are approved for:
- Individuals aged 65 and above, who are at greater risks from serious infections.
- Younger adults with certain pre-existing medical conditions that put them at risk for complications.
- Children: The Moderna vaccine is approved for those older than six months, Pfizer for those over five years, and Novavax for those over 12.
Why Are There Limits On COVID-19 Vaccination?
The federal officials claim that broad immunity and few cases of severe illness make the narrowing of eligibility necessary. However, most experts challenge the FDA's estimate of risks vs. benefits. The Center for Infectious Disease Research and Policy at the University of Minnesota's Director, Dr. Michael Osterholm, called the new policy "a disaster waiting to happen," saying it could lower vaccination coverage among those still vulnerable.
Also Read: Michael Clarke Reveals Skin Cancer Surgery, Sparking Urgent Reminder: How To Spot The Signs Early?
qualifying risk factors would be chronic conditions like asthma, diabetes, high blood pressure, depression, compromised immune systems, and a sedentary lifestyle. This inclusively broadened category continues to exclude many healthy individuals despite the fact that they might wish for protection from infection or long COVID.
Vaccination Recommendations for Children and Pregnant Women
The FDA's new paradigm has generated uncertainty about maternal and pediatric vaccination. Although pregnant women and young children continue to be at great risk of severe COVID, both the CDC and FDA have stepped back from routine recommendations. Newborns, who depend on maternal antibodies, are especially exposed. Experts stress that immunizing pregnant women is still the best way to keep babies safe, a subtlety not universally included in present guidance.
Access and Practical Difficulties
Even those who are covered may find impediments to vaccination. Coverage could become spotty, with some policies paying for injections and others not. Doctors may be reluctant to prescribe for vaccines beyond official FDA approval, and in certain states, pharmacists have a legal mandate to follow CDC guidelines, limiting their ability to administer injections. Out-of-pocket cost of vaccination could be over $200, while appointment is still an obstacle.
These concerns instill fears of unequal access, especially among willing-to-be-immunized groups and vulnerable loved ones. The operational barriers might keep many Americans from being vaccinated, further complicating the public health undertaking.
Public Health Repercussions
The U.S. is now seeing a summer wave of COVID, with increasing hospitalizations in kids and over 100 weekly fatalities. Lower availability of vaccines has the potential to worsen such outcomes, say experts. Infectious Disease Society of America President Dr. Tina Tan cautions that restricted access to vaccines has the potential to increase hospitalizations and fatalities, especially in vulnerable populations.
The move also occurs during a period of already lagging vaccination rates in the U.S.: about 23% of adults and 13% of those aged under 18 have received COVID boosters recently. Many parents and young people themselves continue to want to vaccinate themselves or their children but are now confronted with financial, logistical, or regulatory barriers.
While the FDA describes its new policy as a balanced, risk-based approach, public health officials fear it could produce gaps in protection. Individuals with moderate risk, healthy children, and young adults who want vaccine shots may be forced to undergo prescriptions, pay cash, or encounter barriers to receive shots.
The months ahead will be critical, particularly with the anticipated winter COVID boom. Policymakers, healthcare workers, and members of the public will need to weigh how to balance not imposing unnecessary vaccines and a guarantee of protection for those still at risk.
The FDA's updated approval of COVID-19 vaccines is a new policy shift in U.S. pandemic policy, restricting access to the most vulnerable and leaving healthy young adults in suspension. With conflicting guidance, reduced funding, and operational complexities, the new system has raised concerns regarding equity, security, and long-term impacts on public health. As the U.S. prepares for seasonal peaks and continued virus evolution, the equilibrium between protection and caution will be watched closely by policymakers, healthcare providers, and families across the country.
COVID Cicada variant: Are Children More At Risk? How To Safeguard?

Credit: iStock
The COVID Cicada variant, currently circulating in 23 countries, is likely to affect children more than the previous variants.
Children have largely escaped severe illness from the SARS-COV-2 virus.
However, the new Cicada variant with around 75 genetic changes in its spike protein is likely to disproportionately affect children, as per an expert, who noted its presence in the UK.
“Some people have done analysis on this, suggesting it may be more prevalent among young children. Children get infections all the time, but this might be something to do with the fact that they have never been exposed to Covid vaccines," Prof Ravindra Gupta, of Cambridge University, who advised the UK government during the pandemic, was quoted as saying to The Mirror.
“So this is something we’re looking at in the lab to try and work out why. The problem with this is that it is an infection that spreads fast. Eventually, it ends up in someone vulnerable," he added.
Key Symptoms
Symptoms seem to be similar to those of other recent variants and include
- sore throat,
- cough,
- congestion,
- fatigue,
- headache
- fever.
Also read: Cicada Variant: Will The Current COVID Vaccine Provide Protection?
What Do The 75 Genetic Changes Mean?
Cicada, a descendant of the Omicron variant that emerged in 2021, was first detected in South Africa in 2024. However, it disappeared soon after, only to emerge early this year.
Prof Gupta told the publication that “the Cicada variant developed the 75 mutations to escape immunity, and it evolved by incubating a single patient for over a year”.
"This 'patient zero' would have been unable to clear the virus due to a compromised immune system," due to HIV or anti-cancer drugs.
Further, he explained that due to 75 mutations to the spike protein, the body’s antibodies will take a longer time to fight the Cicada variant.
However, there is no evidence yet that it is likely to cause more severe disease.
Will The Cicada Variant Cause Severe Illness?
The Cicada variant is particularly concerning as it provides no immune protection to people with previous infection or even vaccination.
Yet, the World Health Organization and health experts advise that existing COVID vaccinations can help prevent severe illness and hospitalization.
"It would appear that all the protections we have from our experience with the virus and with vaccines probably offer more limited—not zero—but more limited protection against this strain," Dr. William Schaffner, professor of infectious diseases at Vanderbilt University Medical Center, was quoted as saying by Time.
Also read: COVID Variant BA.3.2 Spreads To 23 Countries: Is The Variant Under Monitoring A Cause Of Worry?
How To Safeguard
Dr. Sai Balasubramanian, a doctor and healthcare strategy executive, writing in Forbes, stressed the need to follow COVID practices such as masks and hand hygiene.
"Healthcare professionals recommend taking general precautions, similar to those used to prevent most viral transmission: get vaccinations when appropriate, wear masks in crowded areas or indoors where there is a high risk of transmission," he said.
He also urged “avoid individuals who have known illness or infections, wash hands frequently, and continue to stay apprised of local community guidelines and the infection spread”.
India To Strictly Inspect GLP-1 Drugs To Curb Misuse: Govt Flags Risks Amid Weight-loss Hype

Credit: iStock
GLP-1 drugs are powerful tools that can improve health, but it comes with risks and caveats. While these are medical therapies, they are being misused as cosmetic products by cosmetologists, physiotherapists, dermatologists, among others.
To curb its misuse and improve public health, the Indian government today issued guidelines on its use, while also flagging risks around it.
In an official statement issued today, the government has stated that it will roll out stricter inspections to curb the misuse of GLP-1 drugs in the country and will punish those who violate the rules.
“GLP-1 drugs are a significant medical breakthrough in treating type 2 diabetes and obesity, but they are not without risk. The drugs carry a wide range of side effects - from common symptoms like nausea and vomiting to severe complications including pancreatitis, kidney injury, and bowel obstruction. These risks make it essential that GLP-1 drugs are only taken under the supervision of registered medical practitioners/specialists,” the statement said.
In the wake of its hype over weight loss, the country's regulatory authorities have taken firm steps to prevent unsupervised use and malpractices in the supply chain.
It has “strongly advised” patients and the public to” consult a qualified medical specialist before use, and to obtain these medications with prescription only through legitimate, regulated channels”.
“Stricter inspections and surveillance will continue in the coming weeks. Businesses that break the rules will face license cancellation, fines, and legal action,” the statement said.
Also read: GLP-1 Drugs: Why India Needs Stricter Rules Now
What Are The Side-effects Of GLP-1 Drugs?
GLP-1 drugs work to keep food in the digestive system for longer – making people feel fuller for longer, thus reducing appetite and inducing weight loss. Hence, the drugs are also prescribed to people with obesity.
However, when done without clinical oversight, it can lead to severe health complications.
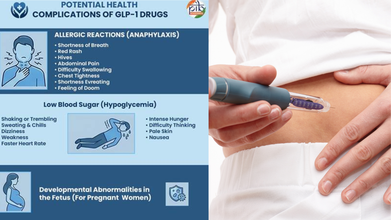
There are various side effects to taking GLP-1 drugs, including nausea and dizziness, to pancreatitis and medullary thyroid cancer.
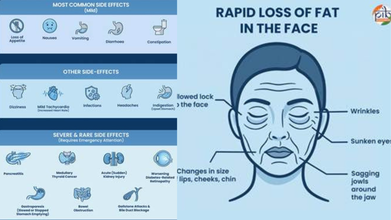
Some serious side effects include
- Pancreatis
- Thyroid Cancer
- Acute (Sudden) Kidney Injury
- Worsening Diabetes-Related Retinopathy
- Gallstone & Bile Duct Blockage
- Rapid loss of fat in the face
- Wrinkles
- Sunken eyes
GLP-1 medications may also complicate various health conditions, such as allergic reactions, with shortness of breath and low blood sugar.
Rush For Generic Weight Loss Drugs
The government's advisory comes as patent for semaglutide -- an active ingredient in diabetes and anti-obesity drugs, specifically Wegovy and Ozempic -- expired on March 20.
This has led to a rush among Indian pharma companies to launch cheaper generic versions, significantly increasing affordability and access for millions battling Type 2 diabetes and obesity.
Also read: CDSCO threatens action against pharma companies for promoting GLP-1 weight-loss drugs
Major Indian drugmakers who launched their generic semglutide injection in the country in March include Sun Pharmaceutical Industries, Zydus Lifesciences, Alkem Laboratories, and Dr. Reddy’s Laboratories.
WHO Guidelines on GLP-1 drugs
Amid the increasing prevalence of GLP-1 drugs, the World Health Organisation (WHO), late last year, acknowledged its role in treating obesity.
However, it warned that medications like GLP-1 alone will not solve the problem affecting more than one billion people worldwide.
The global health body also issued conditional recommendations for using these therapies as part of a comprehensive approach that includes healthy diets, regular physical activity, and support from health professionals.
Ozempic And Wegovy Prices Slashed Again In India To Counter Cheaper Generics

Credit: iStock
Danish pharma giant Novo Nordisk has once again reduced the prices of its blockbuster drugs Ozempic and Wegovy in India. The strategic move is aimed at staying ahead of the entry of the low-cost generic competition in the country.
Starting today, the Ozempic price in India will be cut by 36 percent, while Wegovy has seen a steeper 48 percent reduction — limiting the base price below Rs 6,000 per month.
While Ozempic is available in three dose strengths of 0.25 mg, 0.5 mg, and 1 mg in India, Wegovy has five dose strengths.
Ozempic's and Wegovy's lowest doses of 0.25 mg will now be priced at Rs 1,415 for a weekly shot from Rs 2,200 and Rs 2,712 earlier, respectively, Novo Nordisk India said in a statement on Tuesday.
The average price reduction across doses is 23.8 percent for Ozempic and 27 percent for Wegovy, it added.
Speaking to the media, Novo Nordisk stated that the price cut will benefit a large section of people with diabetes and obesity in India.
The generic weight loss drugs will open up a huge customer base, especially in India, with a high burden of diabetes and obesity.
Novo first slashed Wegovy's price for the first time by up to 37 percent from its launch price last year.
In addition to the entry of generics, the launch of rival Eli Lilly's blockbuster diabetes and obesity drug Mounjaro in India in 2025.
Also read: Semaglutide Becomes Cheap In India: A Gamechanger Or Health Gamble?
Entry Of Generics In India
Soon after the end of Novo Nordisk's patent on semaglutide, on March 20, a host of companies in India launched generic versions of Ozempic.
This includes NATCO Pharma's Semanat and Semafull, priced at Rs 1,290 per month and Rs 1,750 — about 90 per cent cheaper than Ozempic, costing Rs 8,800 per month.
Eris Lifesciences also announced its plans to launch a multi-dose vial under the brand name Sundae at the same price.
Dr. Reddy’s Laboratories, Sun Pharmaceutical Industries, and Glenmark Pharmaceuticals have also launched their generic versions of semaglutide.
Dr. Reddy’s Obeda is priced at Rs 4,200 per month, available in 2 mg and 4 mg strengths.
Sun Pharmaceutical Industries will sell under the brand names Noveltreat (doses ranging from Rs 900) and Sematrinity (doses ranging from Rs 750). Meanwhile, Glenmark’s GLIPIQ vials range from Rs 325 to Rs 440 per week.
Zydus Lifesciences has also announced the launch of its generic version of semaglutide injection under the brand names SEMAGLYNTM, MASHEMATM, and ALTERMET. The average monthly cost of the treatment will be about Rs 2,200, the company said in a statement.
How Does Semaglutide Work?
Semaglutide works as a GLP-1 receptor agonist that mimics the GLP-1 hormone to regulate appetite and blood sugar. It slows gastric emptying and makes you feel fuller longer. It also signals the brain to reduce hunger and cravings, and triggers the pancreas to release insulin when blood sugar is high.
Also read: This Pill Is More Effective For Weight Loss Than Ozempic, Scientists Say
They work by increasing insulin release in a glucose-dependent manner, decreasing the liver's production of glucagon, and slowing down the emptying of the stomach, which helps lower blood sugar levels after a meal. They also act on the brain to suppress appetite and increase feelings of fullness, leading to reduced calorie intake.
In people with type 2 diabetes, notes Harvard Health, the body's cells are resistant to the effects of insulin and the body does not produce enough insulin, or both. This is when GLP-1 agonists stimulate the pancreas to release insulin and suppress the release of another hormone called glucagon.
These drugs also act in the brain to reduce hunger and act on the stomach to delay emptying, so you feel full for a longer time. These effects can lead to weight loss, which can be an important part of managing diabetes.
© 2024 Bennett, Coleman & Company Limited

